Abstract
Five coronaviruses (CoVs) were detected in diarrheal feces from four zoo ruminant species: one wisent (Bison bonasus), two Himalayan tahr (Hemitragus jemlahicus), one sitatunga (Tragelaphus spekii), and one nyala (Tragelaphus angasii). We sequenced and analyzed the spike (S) and hemagglutinin/esterase (HE) genes of these viruses and compared the nucleotide (nt) and deduced amino acid (aa) sequences with those of other bovine CoV (BcoV) strains. Comparison of the entire deduced aa sequences of the S and HE glycoproteins revealed no specific differences that would account for discrimination between bovine-like CoV strains from zoo ruminants and BcoVs strains. In addition, the 99.9% aa identity among the five CoV strains revealed that the ruminants were infected by the same strain. Phylogenetically, bovine-like CoVs belong to group 2a CoVs, which are related most closely to the BcoV strains recently isolated in Korea. These data suggest that cattle are potential reservoirs for CoVs that are capable of infecting zoo ruminants.
Keywords: Bovine-like coronavirus, Bovine coronavirus, Spike (S) gene, Hemagglutinin/esterase (HE) gene, Phylogenetic analysis
1. Introduction
Coronaviruses (CoVs) are a member of the order Nidovirales and family Coronaviridae, each member of which contains a single-stranded, non-segmented RNA positive-sense genome that is 26–31 kb in size. CoVs are classified into three different genetic and antigenic genera, each of which is divided into several subgenera. Bovine-like CoVs and bovine CoVs (BCoVs), which belong to Betacoronavirus 1, contain five major structural proteins: nucleocapsid (N), transmembrane (M), spike (S), small membrane (E), and hemagglutinin/esterase (HE) (Lai and Cavanagh, 1997). Betacoronavirus 1 also include the human CoVs-OC43 (HCoV-OC43), porcine hemagglutinating encephalomyelitis virus (PHEV), equine coronavirus (ECoV), canine respiratory coronavirus (CRCoV) and bubaline coronavirus (BuCoV) (Decaro et al., 2008, Erles et al., 2007, Guy et al., 2000, Haring and Perlman, 2001, Vijgen et al., 2006).
Bovine-like CoVs were first reported in ruminant species in the late 1970s (Tzipori et al., 1978). Later, bovine-like CoVs were also identified in wild animals (sitatunga, waterbuck, musk oxen, llamas, alpacas, white-tailed deer, giraffe, Sambar deer, and sable antelope) (Cebra et al., 2003, Chasey et al., 1984, Hasoksuz et al., 2007, Tsumemitsu et al., 1995). BcoVs cause newborn calf diarrhea (CD), winter dysentery (WD) in adult cattle, and respiratory tract illness in calves and feedlot cattle (Lathrop et al., 2000, Saif et al., 1991, Snodgrass et al., 1986). BcoVs also infect the respiratory tract of cattle and cause disease therein (Cho et al., 2001, Hasoksuz et al., 1999).
The genome of Betacoronavirus 1 encodes a short, spike-like protein known as HE. BcoV uses N-acetyl-9-O-acetylneuraminic acid (sialic acid) as a receptor to initiate infection (Schultze and Herrler, 1992). Although S protein has an affinity for 9-O-acetylated sialic acid, HE protein has been identified as the principal BcoV sialic acid-binding protein (Schultze et al., 1991a, Schultze et al., 1991b). Therefore, HE may be involved in virus entry and/or virus release from infected cells (Schultze et al., 1991a). The S protein, which is composed of two subunits, S1 and S2, has several important functions during the virus–host interaction. The S1 subunit is associated with binding to host cell receptors, whereas the S2 subunit is the viral M protein that mediates the fusion of viral and cellular membranes. Thus, S protein is critical for virus entry and pathogenesis (Schultze et al., 1991b). The variation in host range and tissue tropism among CoV is largely attributable to variations in the S protein (Gallagher and Buchmeier, 2001).
Several Korean BcoV strains have recently been characterized at the molecular level (Jeong et al., 2005a, Jeong et al., 2005b, Park et al., 2006). However, such analyses of bovine-like CoV strains circulating in Korea have not yet been conducted. In this study, we report sequence and phylogenetic analyses of the S and HE protein genes of five bovine-like CoVs isolated from four species of zoo ruminants.
2. Materials and methods
2.1. Clinical signs
The outbreak began with severe diarrhea in a wisent (Bison bonasus) and then spread to other ruminants in the National Zoo during the 2010 winter dysentery (WD) outbreak. Most zoo ruminants presented, apart from the bloody diarrhea, with weakness, depression, anorexia, and dehydration. Sitatunga (Tragelaphus spekii) showed both severe diarrhea and respiratory symptoms. Out of 16 affected animals, a total of 12 fecal samples were collected from four wisents (B. bonasus), three Himalayan tahr (Hemitragus jemlahicus), three sitatunga (T. spekii), and two nyala (Tragelaphus angasii) using rectal swabs. Clinical signs were observed in adult ruminants (more than 24 months of age). No cattle farms were present in the vicinity of the outbreak and no cattle were housed in the Zoo. All fecal samples were examined for common bacterial enteric pathogens (Salmonella spp., Clostridium spp., Campylobacter spp., Mycobacterium spp.) and parasites (Coccidium spp., Crytosporidium spp.). All fecal samples were also analyzed for the presence of BcoV, bovine rotavirus and pestiviruses by RT-PCR and electron microscopy (EM). No enteric pathogens excepting BcoV were detected and isolated in all fecal samples.
2.2. RNA extraction and RT-PCR
Total RNA was extracted from clarified fecal samples using an RNeasy minikit (Qiagen, Valencia, CA) according to the manufacturer's directions. One-step RT-PCR reactions were performed as previously described (Jeong et al., 2005b, Park et al., 2006).
2.3. Sequence determination
PCR products were purified using an agarose gel DNA extraction kit (iNtRON Biotechnology Inc., Daejeon, Korea) and subcloned into the pGEM-T vector (Promega Corp., Madison, WI), according to the manufacturer's directions. Automated nucleotide sequencing of the 5′-UTR gene, inserted into the vector, was performed using a genetic analyzer and cycle sequencing kit (Applied Biosystems, Foster City, CA). All nucleotide positions were confirmed by three or more independent sequencing reactions in both directions.
2.4. Sequence comparison and phylogenetic analysis
Nucleotide sequence alignments were generated using the BioEdit computer program (Ibis Biosciences Inc., Carlsbad, CA). For phylogenetic analyses, complete sequences of the S and HE genes from four zoo ruminants were aligned with reference sequences retrieved from GenBank (Table 1 ). A phylogenetic analysis based on the S and HE sequences was also conducted using BioEdit with the Molecular Evolutionary Genetics Analysis (MEGA) 3.1 program, with bootstrap values based on 3000 replicates. The cutoff point for bootstrap replication was >70%.
Table 1.
GenBank accession numbers of Betacoronavirus 1 reference strains used in the phylogenetic analysis.
| Strain | Year | Host | Clinical forms | Country | GenBank accession no. |
|
|---|---|---|---|---|---|---|
| S | HE | |||||
| Wisent/2010 | 2010 | Wisent | Winter dysentery | Korea | HM573326 | HM573321 |
| Sitatunga/2010 | 2010 | Sitatunga | Winter dysentery | Korea | HM573329 | HM573324 |
| Nyala/2010 | 2010 | Nyala | Winter dysentery | Korea | HM573330 | HM573325 |
| Himalayan Tahr 1/2010 | 2010 | Himalayan tahr | Winter dysentery | Korea | HM573327 | HM573322 |
| Himalayan Tahr 2/2010 | 2010 | Himalayan tahr | Winter dysentery | Korea | HM573328 | HM573323 |
| 0501/2005 | 2005 | Cattle | – | Korea | EU686689 | EU401975 |
| 0502/2005 | 2005 | Cattle | – | Korea | EU401986 | EU401676 |
| BC94/1994 | 1994 | Cattle | – | Korea | DQ389641 | EU401979 |
| SUN5/1994 | 1994 | Cattle | – | Korea | DQ389657 | EU401978 |
| A3/1994 | 1994 | Cattle | – | Korea | AY935640 | EU401977 |
| KCD1/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389632 | DQ389642 |
| KCD2/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389633 | DQ389643 |
| KCD3/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389634 | DQ389644 |
| KCD4/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389635 | DQ389645 |
| KCD5/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389636 | DQ389646 |
| KCD6/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389637 | DQ389647 |
| KCD7/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389638 | DQ389648 |
| KCD8/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389639 | DQ389649 |
| KCD9/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389640 | DQ389650 |
| KCD10/2004 | 2004 | Calf | Calf diarrhea | Korea | DQ389641 | DQ389651 |
| KWD1/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935637 | DQ016118 |
| KWD2/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935638 | DQ016119 |
| KWD3/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935639 | DQ016120 |
| KWD4/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935640 | DQ016121 |
| KWD5/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935641 | DQ016122 |
| KWD6/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935642 | DQ016123 |
| KWD7/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935643 | DQ016124 |
| KWD8/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935644 | DQ016125 |
| KWD9/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935645 | DQ016126 |
| KWD10/2002 | 2002 | Cattle | Winter dysentery | Korea | AY935646 | DQ016127 |
| KWD11/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ389652 | DQ994162 |
| KWD12/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ389653 | DQ994163 |
| KWD13/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994164 | DQ389654 |
| KWD14/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994165 | DQ389655 |
| KWD15/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994166 | DQ389656 |
| KWD16/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994167 | DQ389657 |
| KWD17/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994168 | DQ389658 |
| KWD18/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994169 | DQ389659 |
| KWD19/2002 | 2002 | Cattle | Winter dysentery | Korea | DQ994170 | DQ389660 |
| White-tailed deer/1994 | 1994 | White-tailed deer | Sporadic diarrhea | USA | FJ425187 | FJ425187 |
| Calf-giraffe/2003 | 2003 | Giraffe | Winter dysentery | USA | EF424623 | EF424623 |
| Alpaca/1998 | 1998 | Alpaca | Severe diarrhea | USA | DQ915164 | DQ915164 |
| LUN/1998 | 1998 | Cattle | Bovine shipping fever | USA | AF391542 | AF391542 |
| ENT/1998 | 1998 | Cattle | Bovine shipping fever | USA | AF391541 | AF391541 |
| Sable antelope/2003 | 2003 | Sable antelope | Diarrhea | USA | EF424621 | EF424621 |
| Sambar deer/1994 | 1994 | Sambar deer | Winter dysentery | USA | FJ425188 | FJ425188 |
| Waterbuck/1994 | 1994 | Waterbuck | Winter dysentery | USA | FJ425186 | FJ425186 |
| LSU/1994 | 1994 | Cattle | Respiratory problem | USA | AF058943 | AF058943 |
| OK/1996 | 1996 | Cattle | Respiratory problem | USA | AF058944 | AF058944 |
| E-DB2/1984 | 1984 | Cattle | – | USA | DQ811784 | DQ811784 |
| LY-381 | 1965 | Cattle | – | USA | AF058942 | AF058942 |
| Nordern vaccine/1991 | 1991 | Cattle | – | USA | M64668 | – |
| L9/1991 | 1991 | Cattle | – | USA | M64667 | M76327 |
| Mebus/1972 | 1972 | Cattle | – | USA | U00735 | U00735 |
| Quebec/1972 | 1972 | Cattle | – | Canada | AF220295 | – |
| Bubalus/2007 | 2007 | Water buffalo | Severe diarrhea | Italy | EU019216 | EU019216 |
3. Results and discussion
Of the 12 fecal samples tested, five (one wisent, two Himalayan tahr, one sitatunga, and one nyala) were positive for CoV. Five fecal samples were used for virus isolation and sequencing. All of the S genes sequenced contained an open reading frame (ORF) of 4092 nucleotides, encoding a predicted protein of 1363 amino acid (aa) residues and molecular weight of approximately 150 kDa. Sequence analysis of the five identified strains showed a 99.9% amino acid identity and a genetic relatedness of 99.4–99.5% to two other Korean isolates, BcoV-0501 and BcoV-0502. Thus, the most recent isolates, BcoV-0501 and BcoV-0502 (identified in 2005), had the highest nucleotide identity with the five bovine-like CoVs, whereas they showed only 97.8–97.9% identities with the BC94/1994 (DQ389641) and Sun5/1994 (DQ389657) strains. In total, 86 nucleotide substitutions were found compared to the Mebus strain, but no insertions or deletions were detected in any sequence. Also, 34 substitutions were found in the complete aa sequence of the viral glycoprotein S.
A phylogenetic analysis using reference strains from all known groups of CoVs demonstrated that the bovine-like CoVs isolated in this study belonged to Betacoronavirus 1 clustering with Korean BcoVs isolates collected after 2004 (Fig. 1a). The bovine-like CoVs isolated were closely related to two Korean strains (BcoV-0501 and BcoV-0502) identified in 2005, suggesting that this virus may have been transmitted from cattle to zoo ruminants. Korean BcoVs strains collected before 2004 were divided into several clusters. A BcoV-white-tailed deer strain isolated in 1994 (Tsumemitsu et al., 1995) formed a cluster together with Korean BcoV-KWD1–10 isolates, with the exception of KWD 7.
Fig. 1.
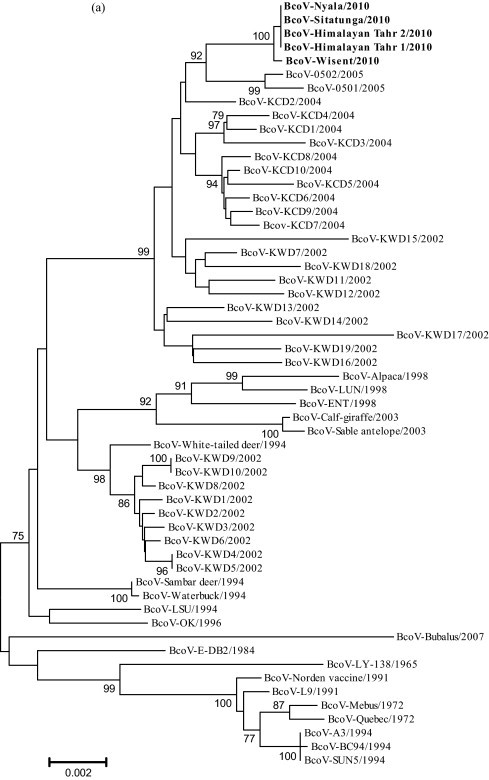
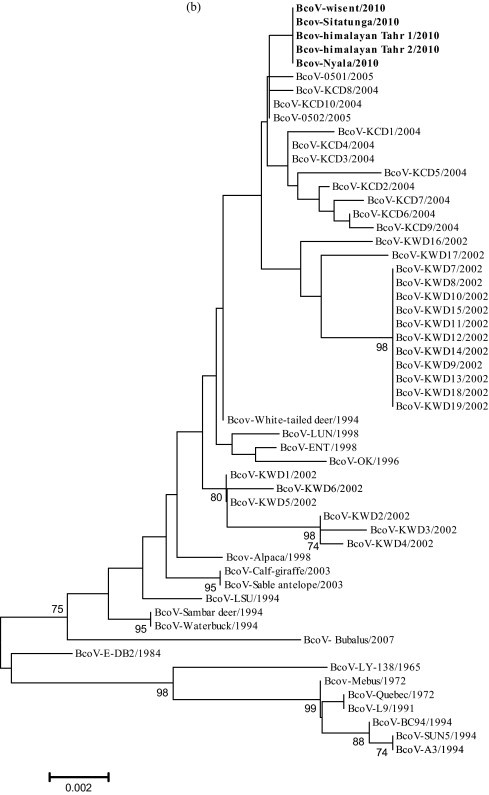
Phylogenetic tree generated by neighbor-joining analysis of genetic distance in the complete sequences of the S (a) and HE (b) genes. The nucleotide sequences of five bovine-like CoV strains were aligned using BioEdit and Mega 3 software. Three thousand bootstrap replicates were subjected to both nucleotide sequence distance and neighbor-joining methods. The consensus phylogenetic tree is shown with bootstrap values >70% displayed above the tree branches.
All of the HE genes contained an ORF of 1272 nucleotides, encoding a predicted protein of 424 aa residues and molecular weight of approximately 47.6 kDa. In total, 27 nucleotide substitutions were found compared to the Mebus strain, but no insertions or deletions were detected in any sequence. Also, five substitutions were found in the complete aa sequence of the S glycoprotein.
Phylogenetic analyses were carried out to determine genetic relationships based on the nucleotide sequences of the HE gene (Fig. 1b). The Korean BcoV strains were divided into three groups. The first group consisted mainly of all of the Korean isolates. CoV strains from zoo ruminants were grouped within a cluster of other Korean CoV strains isolated after 2004. The CoV strains clustered with the BcoV-0501/2005 strain, which was identified in 2005. The second group included some Korean strains isolated in 2002 and bovine-like strains from wild ruminants. The last group consisted mainly of BcoV vaccine strains.
In this study, we compared the S and HE genes of Korean bovine-like CoVs isolated directly from feces of zoo ruminants to determine their relatedness to bovine-like CoVs from wild ruminants at other locations. However, no relationship between Korean bovine-like CoVs and those from wild ruminants at other locations was detected. Sequence analysis of the S and HE genes, however, indicated that the strains show a high similarity to BcoV. This was confirmed by phylogenetic analysis, thus supporting data obtained previously (Alekseev et al., 2008). The CoV strain isolated in this study appears to derive from the same ancestor as the BcoV 05/01 and BcoV 05/02 strains isolated from cattle in Korea. Also, the sequences of the S and HE genes of Korean CoV strains represented on the tree grouped according to year of isolation. These data support the hypothesis that genetic difference may be related more to the time of the appearance of an outbreak than to its geographic location (Alekseev et al., 2008, Jeong et al., 2005b).
The 99.9% aa identity of the five bovine-like CoV strains suggests that the zoo ruminants were infected by the same bovine-like CoV. The high genetic similarity with bovine-like CoVs from the zoo ruminants and Korean BcoV strains was also confirmed, suggesting that cattle may be potential reservoirs for CoVs that infect zoo ruminants. However, additional isolation and characterization of bovine-like CoVs from other heterologous hosts are required to clarify the origin of bovine-like CoVs and their relationship to other BcoVs.
Since the 1970s, bovine-like CoVs have been recognized as being distributed worldwide, with many reports indicating that the virus had been detected and/or isolated in the United States, Europe, and Asia. BcoVs have been detected in the feces of calves and adult cattle with diarrhea in Korea (Jeong et al., 2005a, Jeong et al., 2005b, Park et al., 2006). In addition, antibodies to BcoV have been demonstrated in Korean native goats (Capra hircus), and 1.0% of Korean native goats were serologically positive for BcoV (Yang et al., 2008). However, this to our knowledge is the only report of detection and characterization of bovine-like CoVs in Korea that has been published to date. No specific genomic characteristics that could account for the host specificity exhibited by bovine-like CoVs were identified by comparative sequence analysis.
Acknowledgment
This study was supported by a grant from the National Veterinary Research and Quarantine Service, Ministry of Agriculture, Anyang, Republic of Korea.
References
- Alekseev K.P., Vlasova A.N., Jung K., Hasoksuz M., Zhang X., Halpin R., Wang S., Ghedin E., Spiro D., Saif L.J. Bovine-like coronaviruses isolated from four species of captive wild ruminants are homologous to bovine coronaviruses, based on complete genomic sequences. J. Virol. 2008;82:12422–12431. doi: 10.1128/JVI.01586-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cebra C.K., Mattson D.E., Baker R.J., Sonn R.J., Dearing P.L. Potential pathogens in feces from unweaned llamas and alpacas with diarrhea. J. Am. Vet. Med. Assoc. 2003;223:1806–1808. doi: 10.2460/javma.2003.223.1806. [DOI] [PubMed] [Google Scholar]
- Chasey D., Reynolds D.J., Bridger J.C., Debney T.G., Scott A.C. Identification of coronaviruses in exotic species of Bovidae. Vet. Rec. 1984;115:602–603. doi: 10.1136/vr.115.23.602. [DOI] [PubMed] [Google Scholar]
- Cho K.O., Hoet A.E., Loerch S.C., Wittum T.E., Saif L.J. Evaluation of concurrent shedding of bovine coronavirus via the respiratory tract and enteric route in feedlot cattle. Am. J. Vet. Res. 2001;62:1436–1441. doi: 10.2460/ajvr.2001.62.1436. [DOI] [PubMed] [Google Scholar]
- Decaro N., Martella V., Elia G., Campolo M., Mari V., Desario C., Lucente M.S., Lorusso A., Greco G., Corrente M., Tempesta M., Buonavoglia C. Biological and genetic analysis of a bovine-like coronavirus isolated from water buffalo (Bubalus bubalis) calves. Virology. 2008;370:213–222. doi: 10.1016/j.virol.2007.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erles K., Shiu K.B., Brownlie J. Isolation and sequence analysis of canine respiratory coronavirus. Virus Res. 2007;124:78–87. doi: 10.1016/j.virusres.2006.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallagher T.M., Buchmeier M.J. Coronavirus spike proteins in viral entry and pathogenesis. Virology. 2001;279:371–374. doi: 10.1006/viro.2000.0757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guy J.S., Breslin J.J., Breuhaus B., Vivrette S., Smith L.G. Characterization of a coronavirus isolated from a diarrheic foal. J. Clin. Microbiol. 2000;38:4523–4526. doi: 10.1128/jcm.38.12.4523-4526.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haring J., Perlman S. Mouse hepatitis virus. Curr. Opin. Microbiol. 2001;4:462–466. doi: 10.1016/S1369-5274(00)00236-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasoksuz M., Lathrop S.L., Gadfield K.L., Saif L.J. Isolation of bovine respiratory coronaviruses from feedlot cattle and comparison of their biological and antigenic properties with bovine enteric coronaviruses. Am. J. Vet. Res. 1999;60:1227–1233. [PubMed] [Google Scholar]
- Hasoksuz M., Alekseev K., Vlasova A., Zhang X., Spiro D., Halpin R., Wang S., Ghedin E., Saif L.J. Biological, antigenic, and full-length genomic characterization of a bovine-like coronavirus isolation from a giraffe. J. Virol. 2007;81:4981–4990. doi: 10.1128/JVI.02361-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong J.H., Kim G.Y., Yoon S.S., Park S.J., Kim Y.J., Sung C.M., Jang O.J., Shin S.S., Koh H.B., Lee B.J., Lee C.Y., Kang M.I., Kim H.J., Park N.Y., Cho K.O. Detection and isolation of winter dysentery bovine coronavirus circulated in Korea during 2002–2004. J. Vet. Med. Sci. 2005;67:187–189. doi: 10.1292/jvms.67.187. [DOI] [PubMed] [Google Scholar]
- Jeong J.H., Kim G.Y., Yoon S.S., Park S.J., Kim Y.J., Sung C.M., Shin S.S., Lee B.J., Kang M.I., Park N.Y., Koh H.B., Cho K.O. Molecular analysis of S gene of spike protein of winter dysentery bovine coronavirus circulated in Korea during 2002–2004. Virus Res. 2005;108:207–212. doi: 10.1016/j.virusres.2004.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai M.M.C., Cavanagh D. The molecular biology of coronaviruses. Adv. Virus Res. 1997;48:1–100. doi: 10.1016/S0065-3527(08)60286-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lathrop S.L., Wittum T.E., Brock K.V., Loerch S.C., Perino L.J., Bingham H.R., McCollum F.T., Saif L.J. Association between infection of the respiratory tract attributable to bovine coronavirus and health and growth performance of cattle in feedlots. Am. J. Vet. Res. 2000;61:1062–1066. doi: 10.2460/ajvr.2000.61.1062. [DOI] [PubMed] [Google Scholar]
- Park S.J., Yoon S.S., Choy H.E., Saif L.J., Park S.H., Kim Y.J., Jeong J.H., Park S.I., Kim H.H., Lee B.J., Cho H.S., Kim S.K., Kang M.I., Cho K.O. Detection and characterization of bovine coronaviruses in fecal specimens of adult cattle with diarrhea during the warmer seasons. J. Clin. Microbiol. 2006;44:3187–3188. doi: 10.1128/JCM.02667-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saif L.J., Brock K.V., Redman D.R., Kohler E.M. Winter dysentery in dairy herds: electron microscopic and serological evidence for an association with coronavirus infection. Vet. Rec. 1991;128:447–449. doi: 10.1136/vr.128.19.447. [DOI] [PubMed] [Google Scholar]
- Schultze B., Herrler G. Bovine coronavirus uses N-acetyl-9-O-acetylneuraminic acid as a receptor determinant to initiate the infection of cultured cells. J. Gen. Virol. 1992;73:901–906. doi: 10.1099/0022-1317-73-4-901. [DOI] [PubMed] [Google Scholar]
- Schultze B., Wahn K., Klenk H.D., Herrier G. Isolated HE-protein from hemagglutinating encephalomyelitis virus and bovine coronavirus has receptor-binding activity. Virology. 1991;180:221–228. doi: 10.1016/0042-6822(91)90026-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultze B., Gross H.J., Brossmer R., Herrler G. The S protein of bovine coronavirus is a hemagglutinin recognizing 9-O-acetylated sialic acid as a receptor determinant. J. Virol. 1991;65:6232–6237. doi: 10.1128/jvi.65.11.6232-6237.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snodgrass D.R., Terzolo H.R., Sherwood D., Campbell I., Menzies J.D., Synge B.A. Aetiology of diarrhea in young calves. Vet. Rec. 1986;119:31–34. doi: 10.1136/vr.119.2.31. [DOI] [PubMed] [Google Scholar]
- Tsumemitsu H., el-Kanawati Z.R., Smith D.R., Reed H.H., Saif L.J. Isolation of coronaviruses antigenically indistinguishable from bovine coronavirus from wild ruminants with diarrhea. J. Clin. Microbiol. 1995;33:3264–3269. doi: 10.1128/jcm.33.12.3264-3269.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzipori S., Smith S.M., Makin T., McCaughan C. Enteric coronavirus-like particles in sheep. Aust. Vet. J. 1978;54:320–321. doi: 10.1111/j.1751-0813.1978.tb02478.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vijgen L., Keyaerts E., Lemey P., Maes P., Van Reeth K., Nauwynck H., Pensaert M., Van Ranst M. Evolutionary history of the closely related group 2 coronaviruses: porcine hemagglutinating encephalomyelitis virus, bovine coronavirus, and human coronavirus OC43. J. Virol. 2006;80:7270–7274. doi: 10.1128/JVI.02675-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang D.K., Hwang I.J., Kweon C.H., Lee K.W., Kang M.L., Lee C.S., Cho K.O. Serosurveillance of viral diseases in Korean native goats (Capra hircus) J. Vet. Med. Sci. 2008;70:977–979. doi: 10.1292/jvms.70.977. [DOI] [PubMed] [Google Scholar]



