Abstract
Signaling through the thrombospondin-1 receptor CD47 broadly limits cell and tissue survival of stress, but the molecular mechanisms are incompletely understood. We now show that loss of CD47 permits sustained proliferation of primary murine endothelial cells, increases asymmetric division, and enables these cells to spontaneously reprogram to form multipotent embryoid body-like clusters. c-Myc, Klf4, Oct4, and Sox2 expression is elevated in CD47-null endothelial cells, in several tissues of CD47- and thrombospondin-1-null mice, and in a human T cell line lacking CD47. CD47 knockdown acutely increases mRNA levels of c-Myc and other stem cell transcription factors in cells and in vivo, whereas CD47 ligation by thrombospondin-1 suppresses c-Myc expression. The inhibitory effects of increasing CD47 levels can be overcome by maintaining c-Myc expression and are absent in cells with dysregulated c-Myc. Thus, CD47 antagonists enable cell self-renewal and reprogramming by overcoming negative regulation of c-Myc and other stem cell transcription factors.
CD47 is a signaling receptor for the secreted matricellular protein thrombospondin-1 and the counter-receptor for signal-regulatory protein-α (SIRPα), which on phagocytic cells recognizes CD47 engagement as a marker of self1,2,3. Mice lacking CD47 or thrombospondin-1 are profoundly resistant to tissue stress associated with ischemia, ischemia/reperfusion, and high dose irradiation2,4,5,6,7. The survival advantage of ischemic CD47- and thrombospondin-1-null tissues is mediated in part by increased nitric oxide/cGMP signaling2. Radioresistance associated with CD47 blockade is cell autonomous and independent of NO signaling8, indicating that additional pro-survival signaling pathways are controlled by CD47.
Engaging CD47 in some cell types triggers programmed cell death3,9. BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3) is a pro-apoptotic BH3 domain protein that interacts with the cytoplasmic tail of CD47 and is implicated in CD47-dependent cell death10. Furthermore, CD47 ligation alters localization of the dynamin-related protein Drp1, which controls mitochondria-dependent death pathways9, and some tissues in CD47-null and thrombospondin-1-null mice show increased mitochondrial numbers and function11. Mitochondrial-dependent cell death pathways involving Bcl-2 are limited by the autophagy regulator beclin-112. We recently found that CD47 signaling limits the induction of beclin-1 and other autophagy-related proteins in irradiated cells, and blocking CD47 in vitro and in vivo thereby increases activation of a protective autophagy response13,14. This autophagy response is necessary for the radioprotective effect of CD47 blockade.
In contrast to the above noted survival advantages of decreased CD47 expression, elevated expression of CD47 confers an indirect survival advantage in vivo. CD47 engages SIRPα on macrophages and prevents phagocytic clearance1,15. Similarly, elevated expression of CD47 on several types of cancer cells has been shown to inhibit their killing by macrophages or NK cells16,17,18. Conversely, CD47 antibodies that block SIRPα binding enhance macrophage-dependent clearance of tumors17,19,20,21, although others have shown that such clearance can occur independent of inhibitory SIRPα signaling22,23,24.
Taken together, these studies indicate two opposing roles for CD47 in cell survival. The cell autonomous advantages of decreased CD47 expression, leading to less inhibitory CD47 signaling, must be balanced against the need to maintain sufficient CD47 levels to prevent phagocytic clearance in vivo. Hematopoietic stem cells exhibit elevated CD47 expression, and high CD47 expression in the stem cell niche was proposed to be important to protect stem cells from innate immune surveillance25. In contrast to this protective function of CD47 in stem cells, we now report that loss of CD47 elevates expression of the stem cell transcription factors Sox2, Klf4, Oct4, and c-Myc in primary murine endothelial cells. Consequently, these cells exhibit increased asymmetric cell division and spontaneously and efficiently form clusters that resemble embryoid bodies (EBs) in serum-free media without requiring feeder cells. These EB-like clusters can readily differentiate into various lineages. c-Myc is a global regulator of gene expression in differentiated and stem cells26 and plays a major role in this inhibitory function of CD47. Re-expression of CD47 in null cells down-regulates c-Myc expression and inhibits cell growth, whereas dysregulation of the c-Myc gene, such as commonly occurs in cancer, enables cells to tolerate high CD47 expression.
Results
Loss of CD47 allows self-renewal and increases c-Myc expression
Primary cells isolated from CD47-null mice exhibit a remarkable advantage in adapting to the stress of tissue culture. Lung endothelial cells isolated from WT C57Bl/6 mice had limited survival and proliferative capacities in primary culture as assessed by reduction of [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) and bromodeoxyuridine (BrdU) incorporation (Fig. 1A, B) and rapidly became senescent upon passage (Supplemental Fig. S1). In contrast, CD47-null lung endothelial cells at first passage showed enhanced plating efficiency and proliferation at several cell densities (Fig. 1A, B). Upon repeated passage, WT primary cells became flattened and vacuolated, whereas CD47-null endothelial cells consistently maintained a well differentiated cobblestone morphology for several months in continuous culture (Supplemental Fig. S1). Continuously proliferating cell lines were reproducibly obtained when primary CD47-null cells were repeatedly passaged but very rarely from WT cultures. The CD47-null cells generally lacked expression of the senescence-associated acidic β-galactosidase marker27 that rapidly appeared in the WT cells (Fig. 1C). High frequency generation of continuously proliferating cell lines was also observed for lung endothelial cells cultured from mice lacking the CD47 ligand thrombospondin-1 (Supplemental Fig. S1).
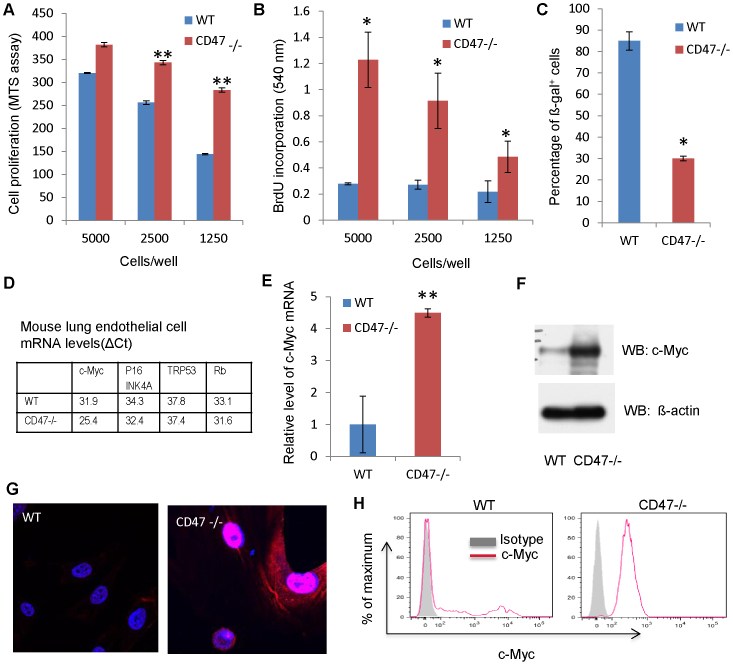
Figure 1. Enhanced proliferation and decreased senescence of CD47-null murine endothelial cells is associated with increased expression of c-Myc.
(A) MTS assay for cell survival and growth over 72 h expressed as % of day 0 values at the indicated plating densities of first passage WT and CD47 null cells. (B) BrdU assay for DNA synthesis. (C) Percentage of senescence-associated β-galactosidase expression at passage 3. (D) Expression of genes associated with cell immortalization in WT and CD47 null cells. (E) c-Myc mRNA levels in lung endothelial cells of WT and CD47 null mice. (F) CD47 limits c-Myc protein levels. (G) c-Myc expression (red) in WT and CD47−/− endothelial cells. Blue = DAPI nuclear stain. (H) Flow cytometric analysis of c-Myc expression in WT and CD47−/− endothelial cells. (*p < 0.05, **p < 0.01).
The ability of CD47-null and thrombospondin-1-null cells to continuously proliferate could reflect either increased escape from senescence or induction of a self-renewing stem cell phenotype. Genes that enable primary cells to escape senescence and become immortalized include p53, Rb, p16-INK4A, and c-Myc28. Of these, only c-Myc mRNA levels were significantly elevated relative to HPRT1 mRNA levels in the primary cell cultures and remained elevated upon repeated passage (Fig. 1D, E). Protein levels for c-Myc were also elevated in primary CD47-null lung endothelial cells as detected by western blotting (Fig. 1F, 2C), immunofluorescence (Fig. 1G) and flow cytometry (Fig. 1H). Most c-Myc was nuclear, but filamentous cytoplasmic staining was also noted in the CD47-null cells, consistent with the known c-Myc association with microtubules29. Characteristic of a pure endothelial cell culture30, the continuously growing CD47-null cells uniformly expressed VEGFR2 and heterogeneously expressed CD31 (Fig. 2D). Absence of vascular smooth muscle cell contamination was indicated by the lack of detectable α-smooth muscle actin expression, although this was detectable at low levels in primary WT endothelial cell cultures (Fig. 2C).
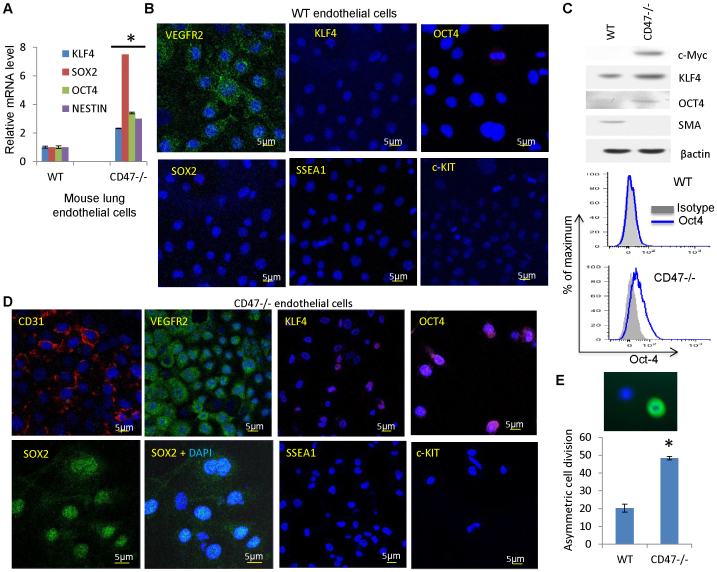
Figure 2. Stem cell and differentiation marker expression in WT and CD47-null endothelial cells.
(A) mRNA expression levels of stem cell transcription factors in WT and CD47 null lung endothelial cells. (B, D) Stem cell and differentiation marker expression in WT (B) and CD47 null mouse endothelial cells (D). The endothelial cells were stained using the indicated antibodies and DAPI (blue). Scale bars = 5 μm. (C) Protein expression of stem cell transcription factors and smooth muscle actin (SMA) assessed by western blotting and flow cytometry (Oct4) in cultured WT or CD47 null endothelial cells in EGM2 medium. (E) Asymmetric cell division frequencies in second passage WT and CD47−/− endothelial cells equilibrium labeled with BrdU and chased for one cell division. Asymmetric division was scored by counting BrdU+(green)/DAPI+ nuclei adjacent to BrdU−/DAPI+ nuclei.
CD47 coordinately regulates stem cell transcription factors
Because elevated c-Myc expression also promotes self-renewal of stem cells31 and is necessary under some conditions for embryonic stem cell self-renewal32, we examined the expression of additional transcription factors that support stem cell reprogramming33 and found significant elevation of mRNA for Sox2, Klf4, and Oct4 and of the stem cell marker nestin in primary CD47-null endothelial cells (Fig. 2A). Oct4 protein expression was detected by immunofluorescence in a majority of the CD47 null cells but not in WT cells (Fig. 2B, D). Sox2 and Klf4 were expressed in a subset of CD47-null cells, whereas Klf4 and c-Myc were undetectable by immunofluorescence in WT endothelial cells, and Sox2 positive cells were rarely seen (Fig. 2B, D). Some Sox2 staining in CD47 null cells was cytoplasmic, consistent with its reported subcellular localization in early embryonic cells34, but the majority of staining was nuclear. Western blotting confirmed elevated levels of Klf4 and Oct4 in CD47-null versus WT cells (Fig. 2C). Flow cytometry also confirmed increased Oct4 expression in CD47-null cells (Fig. 2C).
These data suggested that CD47-null endothelial cell cultures contain a larger fraction of stem cells, which characteristically exhibit asymmetric cell division35,36. We examined the frequency of asymmetric division in WT and CD47-null endothelial cell cultures uniformly labeled using BrdU by chasing with unlabeled medium for 24 h in the presence of cytochalasin B to inhibit cytokinesis. Asymmetric division was indicated by adjacent DAPI+ nuclei where only one cell retained BrdU staining (Fig. 2E upper panel). Such cells were significantly more abundant in the CD47-null cultures (Fig. 2E lower panel).
Efficient formation of embryoid body-like clusters by CD47-null cells
Ectopic expression of Klf4, Sox2, Oct4, and c-Myc or ALK2 bearing an activating mutation in human umbilical vein endothelial cells enables efficiently generation of multipotent or induced pluripotent stem (iPS) cells37,38, suggesting that self-renewal of the CD47-null endothelial cells might arise from increased numbers of stem cells in these cultures. Continuously propagated CD47-null endothelial cells expressed Sca-1, and 24% of the cells were CD14+/CD11+ (Fig. S1). These are characteristic markers for endothelial precursor cells39. However, CD47-null endothelial cells did not express detectable levels of pluripotency marker SSEA-1 or the stem cell marker c-Kit (Fig. 2D). Because elevated expression of c-Myc, Sox2, Oct4 and Klf4 in some types of primary cells is sufficient to induce cystic EB formation40, we examined whether loss of CD47 circumvents the need to artificially elevate these factors for inducing EBs. Indeed, transfer of early passage CD47-null endothelial cells, as well as cultures grown continuously for up to 6 months, into serum-free medium in the absence of any feeder cells within 2 days induced efficient formation of floating cell aggregates that resembled EBs and continued to proliferate in this state (Fig. 3A, Supplemental Fig. S2). These were never observed when primary WT endothelial cells were placed into the same medium, and WT cells did not survive in serum-free media. Cells in the EB-like clusters exhibited strong nuclear c-Myc staining by immunofluorescence (Fig. 3A) and flow cytometry indicated a subpopulation with stronger c-Myc expression than that observed in primary CD47-null endothelial cultures (compare Fig. 3B and Fig. 1H). Unlike CD47-null cells in endothelial cell medium, cells in the EB-like clusters expressed additional stem cell markers including alkaline phosphatase, nestin, SSEA-1 and c-Kit (Fig. 3A, S4). Consistent with their expression of stem cell markers, cells in EB-like clusters frequently exhibited asymmetric cell division (Fig. 3C).
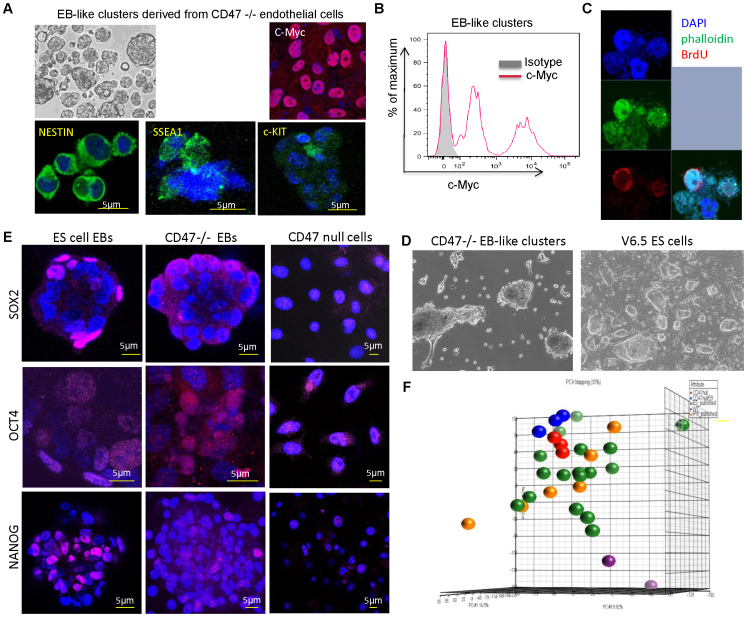
Figure 3. Stem cell marker expression in CD47 null EB-like clusters.
(A) Top left: Typical appearance of EB-like clusters photographed under phase contrast. Top right: Staining with c-Myc antibody (red) and DAPI (blue). Bottom panels: EB-like clusters or cells dissociated from them were stained using the indicated antibodies (green) and DAPI (blue). Scale bars = 5 μm. (B) Flow cytometric analysis of c-Myc expression in CD47-null cells dissociated from EB-like clusters. (C) Detection of asymmetric cell division in cells from CD47-null EB-like clusters equilibrium labeled with BrdU and then chased for two cell divisions without BrdU and stained with anti-BrdU (red) and phalloidin to visualize actin (green). Blue = DAPI. (D) Morphologies of CD47 null EB-like clusters and V6.5 ES cells growing in ES medium with LIF. The V6.5 culture also contains a MEF feeder layer. (E) CD47 null EB-like clusters and V6.5 ES cells cultured as in D and CD47 null endothelial cells in endothelial growth medium (right panels) were stained using the indicated antibodies (magenta) and DAPI (blue). Scale bars = 5 μm. (F) Principal component analysis of global gene expression data for WT (purple) and CD47 null endothelial cells (red), and CD47 null EB-like clusters (blue) compared with published expression data for iPS (gold) and ES cells (green).
CD47−/− cells were grown without feeder cells in ES medium containing LIF adopted colony morphologies similar to V6.5 mouse ES cells grown in the same medium with MEF feeder cells (Fig. 3D). Immunohistochemical analysis of these cells in ES medium demonstrated that CD47 null and V6.5 ES cells contained similar subpopulations that expressed elevated levels of nuclear Oct 4, Sox2, and Nanog (Fig. 3E).
We performed microarray analyses (GSE43133) to globally assess the stem cell character of CD47 null endothelial cells and EB-like clusters derived by culturing in serum free medium for 36 h. A global principal component analysis revealed that CD47 null endothelial cells and EB-like clusters derived from them clustered near published iPS and ES cells, whereas WT endothelial cells did not (Fig. 3F). To characterize the changes in gene expression that accompany EB-like cluster formation in the CD47 null cells, we compared global gene expression in CD47 null EB-like clusters to that of CD47 null cells before removal of serum and endothelial growth factors and found 383 genes with significant changes. Of these 255 clustered with genes that showed similar up or down regulation in the V6.5 ES cells (Supplemental Fig. S3). A GeneSet gene enrichment analysis (GSEA) identified 39 of these up-regulated genes that are included in the molecular signature for human ES cells defined by Bhattacharya41 (Supplemental Fig. S3). The remaining genes included endothelial-specific genes that were significantly down-regulated (e.g. thrombomodulin (Thbd)) and epithelial/mesenchymal transition genes that were induced when CD47 null cells formed EB-like clusters. Notably expression of Kdr, which encodes VEGFR2, decreased 10-fold, consistent with loss of VEGFR2 immunoreactivity in the CD47 null EB-like clusters.
CD47-null EB-like clusters are multipotent
To determine whether CD47 null EB-like clusters are competent to give rise to the three germ layers, 6 day old undifferentiated EB-like clusters prepared from continuously cultured CD47-null endothelial cells were plated on gelatin-coated WillCo dishes for 36 h containing neural, smooth muscle, or hepatocyte differentiation medium (Fig. 4A–C and Movies S1–3). Appearance of cells expressing smooth muscle actin indicated mesoderm differentiation. Cells expressing neuron-specific β III tubulin and glial fibrillary acidic protein (GFAP) indicated ectoderm differentiation. Cells expressing α-fetoprotein (AFP) indicted endoderm differentiation. Because each lineage could arise from different lineage-committed stem cells in the EB-like clusters, we expanded a single clone from CD47-null EB-like clusters and repeated the above differentiation study (Fig. 4D–F). Again, cells expressing characteristic markers of the three embryonic lineages were obtained. Therefore, the CD47-null cells are multipotent.
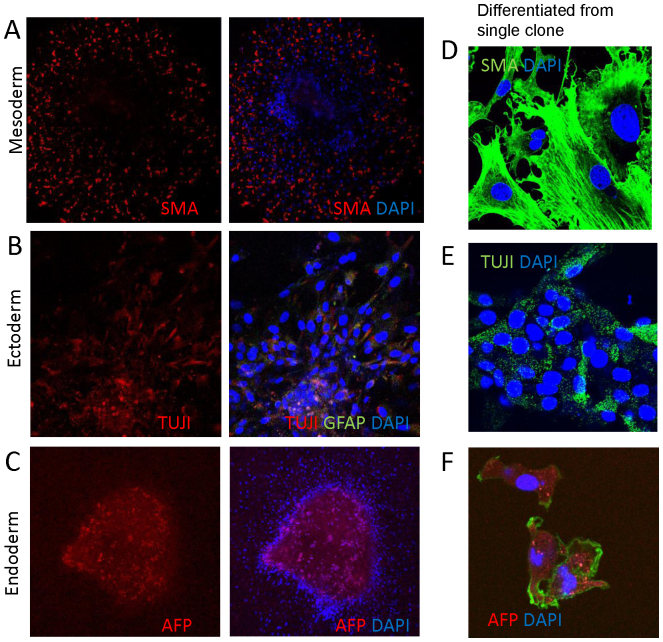
Figure 4. Differentiation of CD47-null EB-like clusters.
(A) EB-like clusters were cultured in the presence of serum for 6 days to induce substrate adhesion and then transferred in to the indicated lineage specific media for 36 h and stained with smooth muscle actin antibody (red) to detect mesodermal cells. (B) Differentiated EB-like clusters were stained with the ectoderm neural markers glial fibrillary acidic protein (GFAP, green), neuron-specific beta III tubulin (TUJI, red). (C) Differentiated EB-like clusters were stained with anti-a-fetoprotein (red) to detect ectodermal cells. In all panels DAPI was used to visualize nuclei (blue). (D–F) A single clone isolated from a CD47-null EB-like cluster in serum-free medium was expanded and then differentiated in the indicated lineage-specific medium for 7 days and stained for the indicated markers. Blue = DAPI, green in (F) = actin.
Morphological differentiation of EBs provides another in vitro assessment of pluripotency42. Differentiation of EB-like clusters in complete RPMI medium for 10–15 days resulted in morphological differentiation characteristic of all three embryonic germ layers (Supplemental Fig. S5). Differentiation was accompanied by loss of SSEA1 expression and decreased expression of other stem cell markers.
These results suggested that CD47 null EB-like clusters contain pluripotent cells. However, we observed limited fibrotic responses and no teratoma formation when CD47 null EB-like clusters were injected into NSG (NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ) mice under conditions where v6.5 ES cells formed teratomas. Therefore, the CD47-null EB-like clusters are probably not fully pluripotent43. Alternatively, teratoma formation may have been prevented by SIRP-dependent clearance of the CD47 null cells15.
Further evidence for multipotency was obtained when undifferentiated CD47-null EB-like clusters were dispersed and cultured in neural medium on a gelatin coated substrate (Supplemental Fig. S6). Ectodermal differentiation was indicated by expression of the neuronal markers MAP2, GFAP, neuron-specific beta III tubulin, and the astrocyte marker S100b respectively (panels d–h). Some nonadherent colonies formed from these cells exhibited extensive neurite formation (panels a–c).
CD47 null endothelial cells cultured in hepatocyte growth medium developed into cystic EB-like clusters and then differentiated into cells that expressed the hepatocyte marker α-fetoprotein (AFP, Supplemental Fig. S6). The CD47-null endothelial cells from which these were derived did not express AFP. Although, a few WT endothelial cells survived in the hepatocyte medium, EB-like clusters never formed, and no expression of AFP was observed.
WT and CD47 null endothelial cells were cultured in mesenchymal cell medium for 10 days. Only CD47 null cells formed EB-like clusters. Some colonies of the mesenchymal differentiated cells exhibited oil red O+ lipid vacuoles characteristic of adipocytes (Supplemental Fig. S6). Dispersion of CD47-null EB-like clusters into serum-free smooth muscle medium containing platelet-derived growth factor and transforming growth factor-β1 resulted in differentiation of cells with typical vascular smooth muscle morphology and expressing the lineage marker smooth muscle actin (Supplemental Fig. S6).
Hematopoietic differentiation of CD47 null endothelial cells
To determine their potential to differentiate into a myeloid lineage, CD47-null endothelial cells were cultured with L929 conditioned medium as a source of macrophage colony stimulating factor (M-CSF)44. After 10 days a change in cell morphology was accompanied by a marked increase in the percentage of Mac2+ cells in treated CD47-null cells compared to the same cells in endothelial growth medium and loss of Sca-1 expression (compare Supplemental Figs. S1 and S7).
In vivo regulation of c-Myc and tissue stem cell abundance by CD47
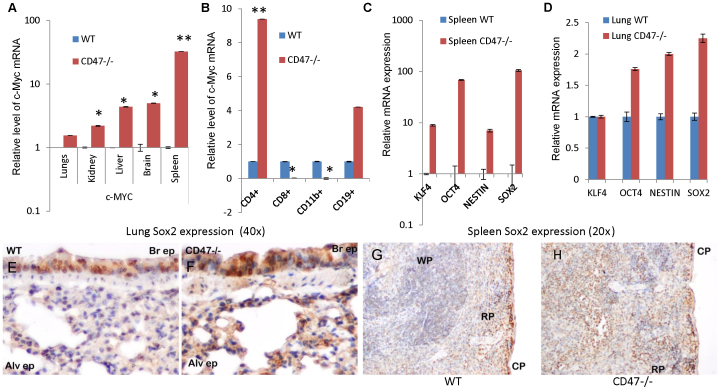
Increased expression of c-Myc mRNA compared to that in WT mice was detected in several organs from CD47-null mice (Fig. 5A). Because the highest elevation occurred in CD47-null spleen, we examined several major cell types from this organ. B (CD19+) and CD4+ T cell populations showed significant up-regulation of c-Myc mRNA, whereas CD8+ T cells and monocytes did not (Fig. 5B). Nestin, Sox2, KLF4, and Oct4 mRNA levels were also markedly elevated in spleen from CD47-null mice (Fig. 5C). Consistent with the lesser elevation of c-Myc mRNA levels in lung, Oct4, Sox2, and nestin mRNA levels were moderately elevated in lung, but KLF4 was not elevated in this organ (Fig. 5D). Sox2 is normally expressed by Clara cells in conducting airways45 and was similarly expressed in WT and CD47-null lung bronchiolar epithelium, but cells expressing higher levels of cytoplasmic Sox2 were selectively observed throughout the alveolar space of the CD47-null lung (Fig. 5E–F).
Figure 5. Loss of CD47 is associated with up-regulation of stem cell transcription factors in vivo.
(A) Relative c-Myc mRNA expression in lung, kidney, liver, brain and spleen of CD47-null versus WT mice. (B) c-Myc mRNA levels in purified splenic cell populations from WT and CD47 null mice. (C) mRNA expression levels in lung from WT and CD47-null mice. (D) mRNA expression levels in spleen from WT and CD47-null mice. (*p < 0.05, **p < 0.01). (E–H) Increased frequency of Sox2 expressing cells in tissues from CD47-null mice. The alveolar (Alv) regions of lung tissues from WT (E) generally lack Sox2-positive cells (brown stain), whereas CD47-null lung shows more positive cells (F). In contrast, similar uniform Sox2 staining was observed in bronchiolar epithelium (BrEp) from WT and null mice, consistent with its previously reported expression in Clara cells45. (G–H) Paraffin embedded sections of representative spleen tissues from WT (G) and CD47−/− (H) mice were stained with a specific antibody to Sox2. Sections were examined under light microscopy showing subcapsular (CP), red pulp (RP) and white pulp (WP) staining.
The spleen of adult mice contains a pool of multipotent CD45−/Hox11+ stem cells that reside in the sub-capsular red pulp and are capable of differentiating into diverse lineages46,47. Consistent with these reports, we observed a limited number of cells with nuclear Sox2 protein expression in the sub-capsular region of WT mouse spleen (Fig. 5G) and sparse expression of Sox2-expressing cells in other compartments of the spleen. Similar subcapsular Sox2 immunoreactivity was seen in spleen sections from CD47−/− mice, but more extensive staining was observed in the adjacent red pulp (Fig. 5H, Supplemental Fig. S7). These differences in Sox2 protein expression are consistent with the whole organ mRNA expression data and suggest that the absence of CD47 increases the number of tissue resident stem cells in vivo.
CD47 expression acutely inhibits c-Myc expression
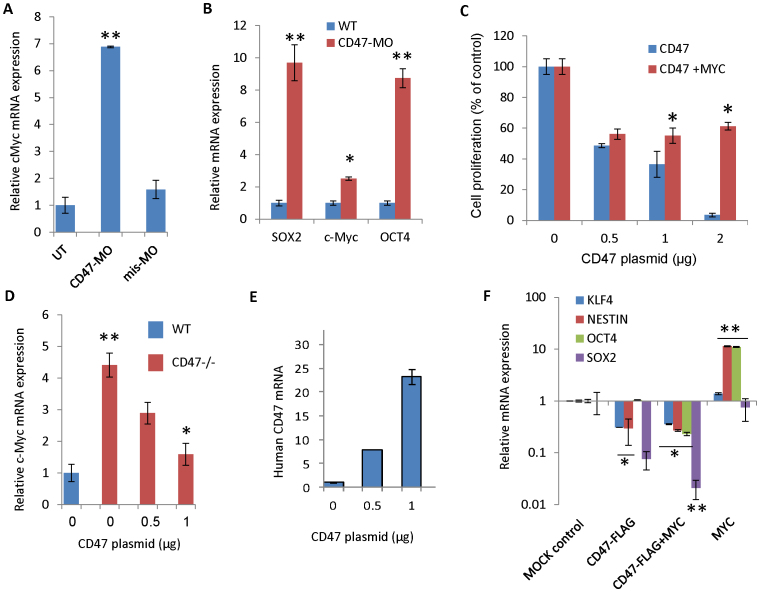
The above results establish a genetic linkage between CD47, maintenance of stem cells, and c-Myc expression but do not prove that CD47 signaling directly controls expression of c-Myc or the other stem cell transcription factors. To clarify this relationship, WT splenic T cells from c-Myc-EGFP knock-in mice26 were treated with a previously validated CD47-targeting antisense morpholino48 and resulted in a 7-fold increase in c-Myc mRNA level at 24 h (Fig. 6A). Intraperitoneal injection of the CD47 morpholino into WT mice significantly decreased CD47 protein expression in vivo (Supplemental Fig. S8) and resulted in induction of c-Myc as well Oct4 and Sox2 mRNA levels in spleen at 48 h (Fig. 6B). These results are consistent with our previous report that intraperitoneal injection of this CD47 morpholino confers radioprotection to mice8.
Figure 6. CD47 expression regulates c-Myc and stem cell transcription factor expression.
(A) Morpholino knockdown of CD47 in lung endothelial cells increases c-Myc mRNA expression. (B) In vivo morpholino knockdown of CD47 elevates c-Myc, Oct4, and Sox2 mRNA at 48 h in mouse spleen. (C) CD47 re-expression in CD47-null murine endothelial cells suppresses cell growth unless c-Myc expression is sustained. (D) CD47 re-expression in CD47 null endothelial cells alters expression c-Myc compared with WT (E) Expression level of transfected human CD47. (F) Re-expression of CD47 and c-Myc alters mRNA expression of stem cell transcription factors. (*p < 0.05, **p < 0.01).
Conversely, re-expression of CD47 in CD47-null endothelial cells by transiently transfecting a human CD47 expression plasmid inhibited their proliferation and viability (Fig. 6C). c-Myc mRNA and protein levels fell when CD47 was re-expressed at a level sufficient to cause growth inhibition (Fig. 6D–E, Supplemental Fig. S8). Growth suppression by transiently expressing CD47 could be partially overcome by co-transfecting the cells with a c-Myc expression vector (Fig. 6C). Transient re-expression of CD47 in the null endothelial cells also decreased mRNA levels for KLF4, Sox2, and nestin (Fig. 6F). Notably, over-expressing c-Myc alone increased nestin and Oct4 mRNA expression, but co-expression of c-Myc with CD47 did not overcome the inhibitory effect of CD47 expression on KLF4, Sox2, or nestin, indicating that these CD47 signaling targets are Myc-independent.
Thrombospondin-1 controls c-Myc via CD47
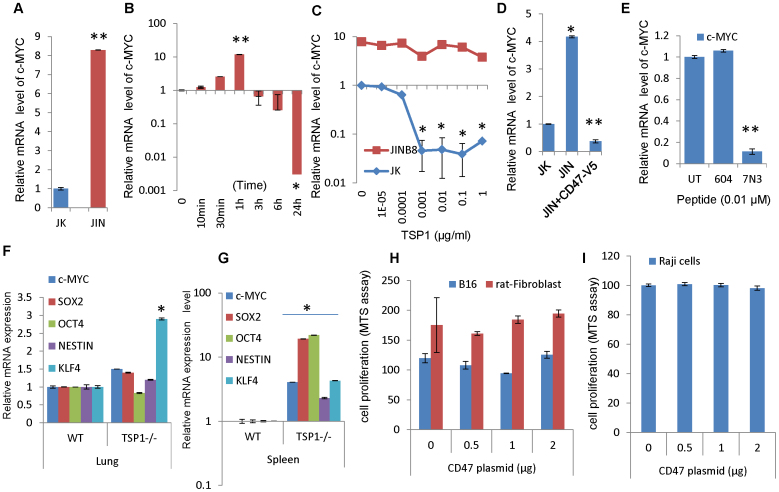
The JinB8 somatic mutant of the Jurkat human T lymphoma cell line lacks CD4749 and exhibited a similar over-expression of c-Myc mRNA relative to the parental Jurkat cells (Fig. 7A). Therefore, CD47 also negatively regulates c-Myc expression in human cells. Mice lacking the CD47 ligand thrombospondin-1 share most of the stress resistance phenotypes of CD47 null mice2,7, and muscle explants from thrombospondin-1-null mice exhibit increased vascular outgrowth into 3-dimensional collagen gels relative to WT explants50. Consistent with these observations and the continuous growth of thrombospondin-1-null endothelial cells, c-Myc levels in Jurkat T cells were transiently induced but then strongly inhibited by treatment with 2.2 nM thrombospondin-1 (Fig. 7B). Picomolar concentrations of thrombospondin-1 were sufficient to inhibit c-Myc expression in Jurkat cells at 24 h, but the elevated c-Myc mRNA levels in Jurkat cells lacking CD47 were not significantly inhibited by thrombospondin-1 (Fig. 7C). Re-expression of CD47 in JinB8 cells reduced c-Myc mRNA expression (Fig. 7D) and restored the ability of thrombospondin-1 to inhibit c-Myc expression (Supplemental Fig. S8). Therefore, CD47 is necessary for this activity of thrombospondin-1. The transient induction of c-Myc by thrombospondin-1 may be mediated by its other receptors expressed by Jurkat cells51.
Figure 7. Regulation of c-Myc and stem cell transcription factors by CD47 ligation.
(A) c-Myc mRNA in Jurkat (JK) and CD47-deficient JinB8 Jurkat T cells (JIN). (B) Time-dependence for regulation of c-Myc mRNA expression by the CD47 ligand thrombospondin-1. Jurkat cells were treated with 2.2 nM thrombospondin-1 for the indicated times before isolating RNA and assessing c-Myc mRNA by real time PCR normalized to β2m mRNA and expressed as a ratio to normalized c-Myc levels in control cells at the corresponding time points. (C) Thrombospondin-1 effects on c-Myc mRNA in WT Jurkat and CD47-deficient (JinB8) T lymphoma cells. (D) CD47 re-expression in JinB8 cells alters expression c-Myc compared with WT Jurkat cells. (E) Effects of CD47-binding TSP1 peptide 7N3 and control peptide 604 on c-Myc mRNA expression in Jurkat T cells. (F and G) mRNA levels in thrombospondin-1-null vs. WT lung and spleen. (H) CD47 over-expression in Rat1 fibroblasts and B16 melanoma cells does not suppress growth. (I) Deregulation of translocated c-Myc in Raji Burkitt's lymphoma cells prevents growth regulation by CD47 over-expression. (*p < 0.05, **p < 0.01).
Similar suppression of c-Myc levels was observed in the presence of a CD47-binding peptide derived from thrombospondin-1 (peptide 7N3, Fig. 7E). A control peptide with a mutated CD47 binding motif (peptide 604) was inactive. Therefore, CD47 engagement is sufficient to inhibit c-Myc expression without the participation of other thrombospondin-1 receptors.
Endogenous thrombospondin-1 also controls expression of c-Myc mRNA in vivo (Fig. 7F, G). c-Myc mRNA levels were elevated approximately 3-fold in thrombospondin-1-null spleen and lung tissues relative to the corresponding WT organs. Consistent with the data for CD47-null mice, Oct4, Sox2 and KLF4 mRNA levels were also elevated in thrombospondin-1-null spleen, but their levels were not significantly increased in lung.
Dysregulation of c-Myc confers resistance to CD47 signaling
One paradox that arises from the above results is that high CD47 expression appears to be a disadvantage for cells because it suppresses c-Myc expression, yet many tumor cells and some stem cells have elevated CD47 expression17,19,20,21. One possible explanation is that other pathways that drive cMyc expression could overcome the inhibitory effects of CD47 signaling. To examine whether c-Myc is the primary target of CD47 signaling that inhibits cell growth, we used Myc-null rat1 fibroblasts that constitutively express a tamoxifen activatable c-Myc-estrogen receptor chimeric protein52. In contrast to cells expressing only native c-Myc controlled by its endogenous promoter, transfecting the Myc-expressing Rat1 fibroblasts with CD47 did not inhibit their growth (Fig. 7H).
We previously reported that blocking CD47 conferred radioprotection to normal cells and mice, but B16 melanomas grown in these mice were not protected and instead showed enhanced radiosensitivity when CD47 was blocked8. This, combined with previous evidence that c-Myc expression is dysregulated in B16 cells53, suggested that CD47 signaling might not regulate c-Myc in these cells. Consistent with this hypothesis, transiently over-expressing CD47 in B16 melanoma cells did not inhibit their growth (Fig. 7H).
Over-expression of CD47 also failed to inhibit growth or survival of Raji Burkitt's lymphoma cells where c-Myc expression is driven by enhancer regions donated by the translocated immunoglobulin heavy chain54 (Fig. 7I). In agreement with these growth data, cell cytoxicity (LDH release) was increased by re-expressing or over expressing CD47-FLAG in normal mouse lung endothelial cells but not in B16 melanoma, Raji Burkitt's lymphoma, and Myc null Rat 1 fibroblasts (Supplemental Fig. S8). Together, these results establish that c-Myc is the dominant target of CD47 signaling for limiting cell growth and suggest that this regulation requires 5′-regions of the c-Myc gene, which are absent in Raji cells.
Discussion
These results demonstrate that a population of multipotent stem cells is selectively maintained at high frequency in primary and continuously cultured CD47-null endothelial cells. These cells support long term maintenance of viable endothelial cells in medium containing endothelial growth factors, but when deprived of serum CD47-null cells spontaneously generate EB-like clusters that express pluripotency markers including alkaline phosphatase, Nanog, and SSEA-1. These in turn can be induced to differentiate into cell types representative of all three embryonic germ layers when appropriate growth factors and cytokines are provided. In contrast to exhibiting these characteristics of iPS cells, the CD47 null EB-like clusters did not form teratomas. Consistent with their lack of teratoma formation, we found no loss of expression of the tumor suppressor gene PTEN or activation of oncogenes including Ras in CD47-null EB-like clusters. Loss of PTEN was reported to increase teratoma formation by pluripotent stem cells55. Others have shown that stem cells can remain pluripotent when teratoma formation is suppressed56. Thus, the CD47-null EB-like clusters may not be fully pluripotent, but their lack of tumorigenicity would be an advantage for potential therapeutic applications57.
Our data reveal that suppression of c-Myc expression is an important mechanism by which thrombospondin-1 signaling via CD47 controls cell growth and differentiation. c-Myc is now recognized to be a universal amplifier of the expression of actively transcribed genes in somatic and embryonic stem cells26, so the ability of CD47 to control c-Myc expression identifies CD47 as cell surface receptor that globally regulates gene expression. Combined with its specific regulation of the stem cell transcription factors Oct4, Sox2 and Klf4, CD47 limits the growth, self-renewal, and reprogramming capacity of primary murine cells in tissue culture. Suppression of these major stem cell transcription factors by CD47 also occurs in vivo and can be modulated by targeting CD47. A corresponding increase in the abundance of tissue stem cells, suggested by the increased numbers of Sox2-positive cells in several organs of CD47-null mice, may contribute to the remarkable ability of tissues in these mice and in thrombospondin-1 null mice to recover from various injuries2,58. In addition to the potential therapeutic utility of CD47 antagonists for treating such injuries, the present data suggest that antagonists of CD47 signaling could be used to increase the ex vivo expansion of tissue stem cells for cell-based therapies and tissue engineering. We propose that CD47 antagonists may also be useful to enhance the generation of lineage-committed or multipotent stem cells and to circumvent the requirement for ectopic expression using plasmids or integrating retroviruses encoding tumor promoting genes such as c-Myc.
c-Myc expression greatly increases the frequency of iPS cells induced by combined ectopic expression of Oct3/4, Sox2, and Klf432. Because our data show that CD47 limits c-Myc expression and other studies have shown that thrombospondin-1 inhibits endothelial progenitor cell function via CD4759, it is remarkable that CD47 expression is elevated on hematopoietic stem cells25. CD47 in this context was proposed to prevent clearance of stem cells by NK cells or macrophages expressing the CD47 counter-receptor SIRPα16,25, but we propose that such stem cells must adapt to the cell-autonomous inhibitory effects of high CD47 expression that suppress c-Myc and other stem cell transcription factors. Myc expression is presumably maintained through other regulatory pathways to preserve viability. The loss of viability that we observe following the acute withdrawal of c-Myc due to CD47-ligation or restoring CD47 expression in null cells may be an example of oncogene addiction in normal cells. These results indicate that a downward excursion of Myc must be carefully managed to prevent cell death or senescence.
Previous studies have implicated thrombospondin-1 as an inhibitor of certain stem cell functions but have not invoked CD47 as the relevant receptor. Thrombospondin-1 null mice exhibited 5-fold more circulating endothelial lineage-committed stem cells (EPCs, CD13+/VEGFR-2+/CD45−/CD117+) than WT mice60. Because the elevation in EPCs was suppressed when the null mice were treated with a drug targeting the thrombospondin receptor CD36, the increased number of EPCs was attributed to loss of anti-angiogenic thrombospondin-1 signaling via CD36 in the null. Conversely, elevated thrombospondin-1 levels in diabetes and peripheral artery disease have been associated with suppression of vascular wound repair mediated by EPCs59,61. Notably, EPCs highly express CD47, and suppression of CD47 by RNAi enhanced their proliferation and angiogenic potential59. The authors attributed this to increased activity of the SDF-1/CXCR4 pathway, but the present data reveal a novel and much broader role of CD47 to limit stem cell function by suppressing c-Myc and other stem cell transcription factors. Furthermore, because CD47 null stem cells show an enhanced capacity to differentiate along diverse lineages, we argue that the inhibitory function of CD47 in stem cell maintenance is not restricted to the endothelial lineage.
These findings add new significance to the previously reported inhibition of thrombospondin-1 transcription and mRNA stability by elevated expression of c-Myc62,63. Combined with our results, thrombospondin-1 and c-Myc can be seen to form a negative feedback loop in normal cells that limits the expression of both genes. This feedback would normally limit the expression of inhibitory thrombospondin-1 and thereby promote tissue renewal and regeneration. This feedback regulation is disrupted in Burkitt's lymphoma where transcriptional control of Myc expression by thrombospondin-1 is lost. Thus, c-Myc dysregulation may allow tumor cells to maintain the elevated surface expression of CD47 that was first identified in ovarian carcinoma64 and subsequently extended to other cancers3,17,18,21,25. Conversely, aging and chronic diseases such as diabetes and pulmonary hypertension where thrombospondin-1 expression is elevated should limit c-Myc expression65,66,67, which could contribute to pathogenesis of these diseases by limiting tissue stem cell maintenance and function.
As an inhibitory cell surface receptor that controls self-renewal, CD47 may be critical for understanding how the microenvironment in the stem cell niche regulates stem cell differentiation68. CD47 may directly transmit a negative signal from the environment that inhibits self-renewal or proliferation, or lateral cross talk of CD47 with integrins and growth factor receptors in the plasma membrane3,69 may negatively modulate these signals in stem cells. Our studies identify thrombospondin-1 as a key environmental signal that inhibits stem cell self-renewal via CD47.
Methods
Animals
Thrombospondin-1 null70 CD47 null mice71 extensively back-crossed onto a C57Bl/6J background and WT mice were housed in a specific pathogen-free environment and had ad libitum access to standard rat chow and water. Care and handling of animals and all experimental procedures used here were approved under protocol LP-012 and in compliance with standards established by the Animal Care and Use Committee of the National Cancer Institute.
Cells
Mouse lung endothelial cells were isolated and their purity verified as described previously50. These conditions were previously documented to reproducibly yield >95% pure endothelial cells at passage 2 (CD31+, smooth muscle actin−). Mouse lung endothelial cells were cultured at 37°C with 5% CO2 using Endothelial Growth Medium-2 (EGM2, Fisher Scientific). Cell populations from mouse spleens were separated using Pan T cell Isolation, CD4(L3T4), CD8a(Ly-2) CD11b, and CD19 microbead kits (Miltenyi Biotech).
V6.5 mouse embryonic stem (ES) cells were cultured on gelatin-coated dishes with mouse embryonic fibroblast (MEF) feeder cells using standard mES medium containing DMEM (high glucose), 15% ES cell-qualified FBS, 200 mM L-glutamine, non-essential amino acids (LifeTechnology), Pen/Strep, 0.1 mM 2-mercaptoethanol, and 1000 unit/ml LIF.
Western blots
Equal numbers of lung endothelial cells from WT and CD47 null were plated in 6-well plates overnight. Cell lysates were made from washed cells using NP-40 lysis buffer (50 mM Tris pH 8.0, 150 mM NaCl and 1% NP-40 along with ProteoBlock Protease inhibitor Cocktail (Fermentas)). The lysates were centrifuged, and equal volumes of supernatant were boiled with 4× NuPAGE–LDS sample buffer (Invitrogen) for 5 min at 95°C. Proteins were separated using 4–12% Bis-Tris gels (Invitrogen). N-terminal c-Myc antibody (Epitomics), human CD47 -B6H12, V5-peroxidase (Invitrogen) and β-actin (Sigma) were used at 1:1000 to perform western blots. Secondary anti-rabbit IgG or anti-mouse IgG conjugated to HRP were used at 1:5000. Super Signal West Pico chemiluminescent substrate (Fisher Scientific) was used to detect bound antibodies. For protein normalization, the blots were reprobed using a β-actin antibody (Sigma Aldrich).
Sox2 immunohistochemistry
Lung and spleen tissues from groups of 3 WT and CD47−/− mice were fixed in 10% formalin. Tissue was paraffin embedded and 5 μm thick sections were immunostained using a Sox2 antibody (1:100) or a non-specific control antibody and detected using the DAKO LASB Universal Kit. Stained sections were visualized and photographed under light microscope using the Q-Imaging system.
Transient CD47 re- or over-expression
Isolated mouse lung endothelial cells, Raji Burkitt's lymphoma cells, B16 melanoma cells, and Myc null Rat1 fibroblast cells were plated overnight in 6-well plates. The cells were transfected with CD47-FLAG expression plasmid72 and/or human c-Myc-GFP plasmid26. The cells were transfected using Opti-MEM® I Reduced Serum Medium (Invitrogen) and the FuGENE HD Transfection kit (Roche). The serum free medium was replaced with complete RPMI medium 5 h after transfection. The cells were analyzed at 24–36 h post transfection. The supernatants were analyzed for lactate dehydrogenase (LDH) release using CytoTox 96® Non-Radioactive Cytotoxicity Assay (Promega).
The JINB8 cells were transfected with CD47-V5 construct using Amaxa (Lonza). The transfection efficiency was determined by flow Cytometry. The CD47-V5 transfected cells were purified using magnetic beads preincubated with human CD47 antibody (B6H12). The transfected cells were loaded onto MACS column which was placed in the magnetic field of a MACS separator. The magnetic bead labeled CD47+ cells were retained on the column. The unbound cells were depleted from CD47+ cells. After removal of the column, the CD47+ cells bound to magnetic beads were eluted. The Pure population of CD47+ was stably cultured using G418 drug (250 ug/ml). The Pure population of CD47+ and Jurkat cells were centrifuged and re-suspended in RPMI + 1% FBS at 106/ml. Cells were plated in 12 well plates and treated with 1 μg/ml (2.2 nM) thrombospondin-1, and total RNA was isolated using TRIzol. The relative gene expression of c-MYC was measured using GAPDH as a control.
CD47 knockdown in T cells and in WT mice
A translation-blocking antisense morpholino oligonucleotide complementary to human and murine CD47 (CGTCACAGGCAGGACCCACTGCCCA) and a 5-base mismatch control morpholino (CGTgACAGcCAcGACCgACTGCgCA) were obtained from GeneTools as previously described48. Primary T cells isolated from c-Myc EGFP knock-in mouse were transfected using morpholinos (2.5 μM) along with Endoporter according to manufacturer's instructions. Mice were treated by injection of 750 μl of 10 μM morpholino in saline as described8. Organs were harvested for mRNA isolation after 48 h.
Modulation of stem cell transcription factors by thrombospondin-1
Jurkat and JinB8 cells were centrifuged and re-suspended in RPMI + 1% FBS at 106/ml. Cells were plated in 12 well plates and treated with 1 μg/ml (2.2 nM) thrombospondin-1 using the indicated times and concentrations, and total RNA was isolated using TRIzol.
Cell proliferation assays
Equal numbers of mouse lung endothelial cells from WT and CD47 (Passage 1) were plated in 96 well plates using RPM1+1% FBS. After 72 h, net cell proliferation was assessed by the increase in formazan absorbance versus controls assessed at time 0 using Cell Titer 96R aqueous MTS kit (Promega). DNA synthesis was measured using a BrdU Assay (EMD Biosciences).
Senescence-associated β-galactosidase assay
Senescent cells were detected in WT and CD47 null lung endothelial cells (Passage 3) by staining for a senescence-associated β-galactosidase73. Positive cell numbers were expressed as a percentage of the total cells.
Statistical analysis
Two-way ANOVA with replication was used for analyzing real time PCR. Student t-test was used for cell proliferation, cell cytotoxicity and cell senescence assays, which were performed in triplicate. All results are presented as mean ± SD.
Author Contributions
S.K., D.S.P., J.S.I., D.L. and D.D.R. conceived and designed the experiments. S.K., D.S.P., E.V.S., C.L., A.G.E., M.L.P., S.P.S., A.N., Z.N. and J.S.I. performed experiments and analyzed data. S.K., D.S.P., E.V.S., C.L. A.G.E., D.L., J.S.I. and D.D.R. wrote the manuscript. All authors reviewed the manuscript.
Supplementary Material
movie S1
movie S2
movie S3
supplementary methods and figures
Acknowledgments
This work was supported by the Intramural Research Programs of the NIH/NCI (D.D.R., D.L.) and NHGRI (A.G.E.), 1RO1HL108954-01 (NIH/NHLBI), 1P01HL103455-01 (NIH), 11BGIA7210001 (AHA), the Institute for Transfusion Medicine, and the Western Pennsylvania Hemophilia Center (J.S.I.). We thank Dr. Yosuke Mukoyama for providing reagents, Judith Horn and Maryann Williamson (Laboratory of Pathology, NCI) for their help for preparing and staining sections, Weiwei Wu for microarray analysis, and Susan Garfield, Poonam Manan and Tonya Huang (NCI) for confocal microscopy.
References
- Matozaki T., Murata Y., Okazawa H. & Ohnishi H. Functions and molecular mechanisms of the CD47-SIRPalpha signalling pathway. Trends Cell Biol 19, 72–80 (2009). [DOI] [PubMed] [Google Scholar]
- Roberts D. D., Miller T. W., Rogers N. M., Yao M. & Isenberg J. S. The matricellular protein thrombospondin-1 globally regulates cardiovascular function and responses to stress. Matrix Biol. 31, 162–169 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frazier W. A., Isenberg J. S., Kaur S. & Roberts D. D. in UCSD Nature Molecule Pages (2010). doi:10.1038/mp.a002870.01.
- Isenberg J. S. et al. Thrombospondin-1 limits ischemic tissue survival by inhibiting nitric oxide-mediated vascular smooth muscle relaxation. Blood 109, 1945–1952 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thakar C. V. et al. Identification of thrombospondin 1 (TSP-1) as a novel mediator of cell injury in kidney ischemia. J Clin Invest 115, 3451–3459 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg J. S. et al. Treatment of ischemia/reperfusion injury by limiting thrombospondin-1/CD47 signaling. Surgery 144, 752–761 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg J. S. et al. Thrombospondin-1 and CD47 limit cell and tissue survival of radiation injury. Am. J. Pathol. 173, 1100–1112 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxhimer J. B. et al. Radioprotection in normal tissue and delayed tumor growth by blockade of CD47 signaling. Sci. Transl. Med. 1, 3ra7 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bras M. et al. Drp1 mediates caspase-independent type III cell death in normal and leukemic cells. Mol Cell Biol 27, 7073–7088 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamy L. et al. Interactions between CD47 and thrombospondin reduce inflammation. J Immunol 178, 5930–5939 (2007). [DOI] [PubMed] [Google Scholar]
- Frazier E. P. et al. Age-dependent regulation of skeletal muscle mitochondria by the thrombospondin-1 receptor CD47. Matrix Biol 30, 154–161 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang R., Zeh H. J., Lotze M. T. & Tang D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18, 571–580 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Pantoja D. R. et al. CD47 deficiency confers cell and tissue radioprotection by activation of autophagy. Autophagy 8, 1628–1642 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Pantoja D. R., Ridnour L. A., Wink D. A. & Roberts D. D. Blockade of CD47 increases survival of mice exposed to lethal total body irradiation. Sci Rep 3, 1038 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldenborg P. A. et al. Role of CD47 as a marker of self on red blood cells. Science 288, 2051–2054 (2000). [DOI] [PubMed] [Google Scholar]
- Kim M. J. et al. Association of CD47 with natural killer cell-mediated cytotoxicity of head-and-neck squamous cell carcinoma lines. Tumour Biol 29, 28–34 (2008). [DOI] [PubMed] [Google Scholar]
- Majeti R. et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell 138, 286–299 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan K. S. et al. Identification, molecular characterization, clinical prognosis, and therapeutic targeting of human bladder tumor-initiating cells. Proc Natl Acad Sci U S A 106, 14016–14021 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao M. P. et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142, 699–713 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao M. P. et al. Therapeutic antibody targeting of CD47 eliminates human acute lymphoblastic leukemia. Cancer Res 71, 1374–1384 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willingham S. B. et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci U S A 109, 6662–6667 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X. W. et al. CD47-signal regulatory protein-alpha (SIRPalpha) interactions form a barrier for antibody-mediated tumor cell destruction. Proc Natl Acad Sci U S A 108, 18342–18347 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto-Pantoja D. R., Miller T. W., Frazier W. A. & Roberts D. D. Inhibitory signaling through signal regulatory protein-alpha is not sufficient to explain the antitumor activities of CD47 antibodies. Proc Natl Acad Sci U S A 109, E2842 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao X. W., Kuijpers T. W. & van den Berg T. K. Is targeting of CD47-SIRPalpha enough for treating hematopoietic malignancy? Blood 119, 4333–4334. [DOI] [PubMed] [Google Scholar]
- Jaiswal S. et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138, 271–285 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie Z. et al. c-Myc Is a Universal Amplifier of Expressed Genes in Lymphocytes and Embryonic Stem Cells. Cell 151, 68–79 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurz D. J., Decary S., Hong Y. & Erusalimsky J. D. Senescence-associated (beta)-galactosidase reflects an increase in lysosomal mass during replicative ageing of human endothelial cells. J Cell Sci 113 (Pt 20), 3613–3622 (2000). [DOI] [PubMed] [Google Scholar]
- Wang C., Lisanti M. P. & Liao D. J. Reviewing once more the c-myc and Ras collaboration: converging at the cyclin D1-CDK4 complex and challenging basic concepts of cancer biology. Cell Cycle 10, 57–67 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexandrova N. et al. The N-terminal domain of c-Myc associates with alpha-tubulin and microtubules in vivo and in vitro. Mol Cell Biol 15, 5188–5195 (1995). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pusztaszeri M. P., Seelentag W. & Bosman F. T. Immunohistochemical expression of endothelial markers CD31, CD34, von Willebrand factor, and Fli-1 in normal human tissues. J Histochem Cytochem 54, 385–395 (2006). [DOI] [PubMed] [Google Scholar]
- Kim K. S. et al. Self-renewal induced efficiently, safely, and effective therapeutically with one regulatable gene in a human somatic progenitor cell. Proc Natl Acad Sci U S A 108, 4876–4881 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varlakhanova N. V. et al. myc maintains embryonic stem cell pluripotency and self-renewal. Differentiation 80, 9–19 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okita K. & Yamanaka S. Induced pluripotent stem cells: opportunities and challenges. Philos Trans R Soc Lond B Biol Sci 366, 2198–2207 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keramari M. et al. Sox2 is essential for formation of trophectoderm in the preimplantation embryo. PLoS One 5, e13952 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sundararaman B. et al. Asymmetric chromatid segregation in cardiac progenitor cells is enhanced by Pim-1 kinase. Circ Res 110, 1169–1173 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pine S. R., Ryan B. M., Varticovski L., Robles A. I. & Harris C. C. Microenvironmental modulation of asymmetric cell division in human lung cancer cells. Proc Natl Acad Sci U S A 107, 2195–2200 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panopoulos A. D. et al. Rapid and highly efficient generation of induced pluripotent stem cells from human umbilical vein endothelial cells. PLoS One 6, e19743 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medici D. et al. Conversion of vascular endothelial cells into multipotent stem-like cells. Nat Med 16, 1400–1406 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehman J., Li J., Orschell C. M. & March K. L. Peripheral blood "endothelial progenitor cells" are derived from monocyte/macrophages and secrete angiogenic growth factors. Circulation 107, 1164–1169 (2003). [DOI] [PubMed] [Google Scholar]
- Itskovitz-Eldor J. et al. Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med 6, 88–95 (2000). [PMC free article] [PubMed] [Google Scholar]
- Bhattacharya B. et al. Gene expression in human embryonic stem cell lines: unique molecular signature. Blood 103, 2956–2964 (2004). [DOI] [PubMed] [Google Scholar]
- Sheridan S. D., Surampudi V. & Rao R. R. Analysis of embryoid bodies derived from human induced pluripotent stem cells as a means to assess pluripotency. Stem Cells Int 2012, 738910 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith K. P., Luong M. X. & Stein G. S. Pluripotency: toward a gold standard for human ES and iPS cells. J Cell Physiol 220, 21–29 (2009). [DOI] [PubMed] [Google Scholar]
- Genovesi E. V. et al. In vitro induction of swine peripheral blood monocyte proliferation by the fibroblast-derived murine hematopoietic growth factor CSF-1. Vet Immunol Immunopathol 23, 223–244 (1989). [DOI] [PubMed] [Google Scholar]
- Tompkins D. H. et al. Sox2 is required for maintenance and differentiation of bronchiolar Clara, ciliated, and goblet cells. PLoS One 4, e8248 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faustman D. L. Regenerative medicine: Stem cell research turns to the spleen. Discov Med 5, 447–449 (2005). [PubMed] [Google Scholar]
- Faustman D. L. & Davis M. Stem cells in the spleen: therapeutic potential for Sjogren's syndrome, type I diabetes, and other disorders. Int J Biochem Cell Biol 42, 1576–1579 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg J. S. et al. Increasing survival of ischemic tissue by targeting CD47. Circ Res 100, 712–720 (2007). [DOI] [PubMed] [Google Scholar]
- Reinhold M. I., Green J. M., Lindberg F. P., Ticchioni M. & Brown E. J. Cell spreading distinguishes the mechanism of augmentation of T cell activation by integrin-associated protein/CD47 and CD28. Int Immunol 11, 707–718 (1999). [DOI] [PubMed] [Google Scholar]
- Zhou L., Isenberg J. S., Cao Z. & Roberts D. D. Type I collagen is a molecular target for inhibition of angiogenesis by endogenous thrombospondin-1. Oncogene 25, 536–545 (2006). [DOI] [PubMed] [Google Scholar]
- Li Z. et al. Interactions of thrombospondins with α4β1 integrin and CD47 differentially modulate T cell behavior. J Cell Biol 157, 509–519 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connell B. C. et al. A large scale genetic analysis of c-Myc-regulated gene expression patterns. J Biol Chem 278, 12563–12573 (2003). [DOI] [PubMed] [Google Scholar]
- Huber K. R. et al. Effect of verapamil on cell cycle transit and c-myc gene expression in normal and malignant murine cells. Br J Cancer 59, 714–718 (1989). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanda K., Hu H. M., Zhang L., Grandchamps J. & Boxer L. M. NF-kappa B activity is required for the deregulation of c-myc expression by the immunoglobulin heavy chain enhancer. J Biol Chem 275, 32338–32346 (2000). [DOI] [PubMed] [Google Scholar]
- Lindgren A. G. et al. Loss of Pten causes tumor initiation following differentiation of murine pluripotent stem cells due to failed repression of Nanog. PLoS One 6, e16478 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazquez-Martin A. et al. Metformin limits the tumourigenicity of iPS cells without affecting their pluripotency. Sci Rep 2, 964 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-David U. & Benvenisty N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat Rev Cancer 11, 268–277 (2011). [DOI] [PubMed] [Google Scholar]
- Hayashi H., Sakai K., Baba H. & Sakai T. Thrombospondin-1 is a novel negative regulator of liver regeneration after partial hepatectomy via TGF-beta1 activation in mice. Hepatology 55, 1562–1573 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smadja D. M. et al. Thrombospondin-1 is a plasmatic marker of peripheral arterial disease that modulates endothelial progenitor cell angiogenic properties. Arterioscler Thromb Vasc Biol 31, 551–559 (2011). [DOI] [PubMed] [Google Scholar]
- Shaked Y. et al. Genetic heterogeneity of the vasculogenic phenotype parallels angiogenesis; Implications for cellular surrogate marker analysis of antiangiogenesis. Cancer Cell 7, 101–111 (2005). [DOI] [PubMed] [Google Scholar]
- Ii M. et al. Endothelial progenitor thrombospondin-1 mediates diabetes-induced delay in reendothelialization following arterial injury. Circ Res 98, 697–704 (2006). [DOI] [PubMed] [Google Scholar]
- Janz A., Sevignani C., Kenyon K., Ngo C. V. & Thomas-Tikhonenko A. Activation of the myc oncoprotein leads to increased turnover of thrombospondin-1 mRNA. Nucleic Acids Res 28, 2268–2275 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L. et al. Silencing of thrombospondin-1 is critical for myc-induced metastatic phenotypes in medulloblastoma. Cancer Res 70, 8199–8210 (2010). [DOI] [PubMed] [Google Scholar]
- Mawby W. J., Holmes C. H., Anstee D. J., Spring F. A. & Tanner M. J. Isolation and characterization of CD47 glycoprotein: a multispanning membrane protein which is the same as integrin-associated protein (IAP) and the ovarian tumour marker OA3. Biochem J 304, 525–530 (1994). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isenberg J. S. et al. Blocking thrombospondin-1/CD47 signaling alleviates deleterious effects of aging on tissue responses to ischemia. Arterioscler Thromb Vasc Biol 27, 2582–2588 (2007). [DOI] [PubMed] [Google Scholar]
- Raman P., Harry C., Weber M., Krukovets I. & Stenina O. I. A novel transcriptional mechanism of cell type-specific regulation of vascular gene expression by glucose. Arterioscler Thromb Vasc Biol 31, 634–642 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bauer P. M. et al. Activated CD47 promotes pulmonary arterial hypertension through targeting caveolin-1. Cardiovasc Res 93, 682–693 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons B. D. & Clevers H. Strategies for homeostatic stem cell self-renewal in adult tissues. Cell 145, 851–862 (2011). [DOI] [PubMed] [Google Scholar]
- Kaur S. et al. Thrombospondin-1 inhibits vascular endothelial growth factor receptor-2 signaling by disrupting its association with CD47. J Biol Chem 285, 38923–38932 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawler J. et al. Thrombospondin-1 is required for normal murine pulmonary homeostasis and its absence causes pneumonia. J Clin Invest 101, 982–992 (1998). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindberg F. P. et al. Decreased resistance to bacterial infection and granulocyte defects in IAP-deficient mice. Science 274, 795–798 (1996). [DOI] [PubMed] [Google Scholar]
- Kaur S. et al. Heparan sulfate modification of the transmembrane receptor CD47 is necessary for inhibition of T cell receptor signaling by thrombospondin-1. J Biol Chem 286, 14991–15002 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debacq-Chainiaux F., Erusalimsky J. D., Campisi J. & Toussaint O. Protocols to detect senescence-associated beta-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat Protoc 4, 1798–1806 (2009). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
movie S1
movie S2
movie S3
supplementary methods and figures