Multiple-micronutrient supplementation for women during pregnancy
- PMID: 28407219
- PMCID: PMC6478115
- DOI: 10.1002/14651858.CD004905.pub5
Multiple-micronutrient supplementation for women during pregnancy
Update in
-
Multiple-micronutrient supplementation for women during pregnancy.Cochrane Database Syst Rev. 2019 Mar 14;3(3):CD004905. doi: 10.1002/14651858.CD004905.pub6. Cochrane Database Syst Rev. 2019. PMID: 30873598 Free PMC article.
Abstract
Background: Multiple-micronutrient (MMN) deficiencies often coexist among women of reproductive age in low- to middle-income countries. They are exacerbated in pregnancy due to the increased demands, leading to potentially adverse effects on the mother and developing fetus. Though supplementation with MMNs has been recommended earlier because of the evidence of impact on pregnancy outcomes, a consensus is yet to be reached regarding the replacement of iron and folic acid supplementation with MMNs. Since the last update of this Cochrane review, evidence from a few large trials has recently been made available, the inclusion of which is critical to inform policy.
Objectives: To evaluate the benefits of oral multiple-micronutrient supplementation during pregnancy on maternal, fetal and infant health outcomes.
Search methods: We searched the Cochrane Pregnancy and Childbirth Group's Trials Register (11 March 2015) and reference lists of retrieved articles and key reviews. We also contacted experts in the field for additional and ongoing trials.
Selection criteria: All prospective randomised controlled trials evaluating MMN supplementation with iron and folic acid during pregnancy and its effects on the pregnancy outcome were eligible, irrespective of language or the publication status of the trials. We included cluster-randomised trials, but quasi-randomised trials were excluded.
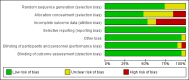
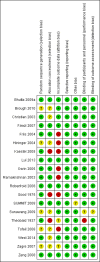
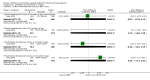
Data collection and analysis: Two review authors independently assessed trials for inclusion and risk of bias, extracted data and checked them for accuracy. The quality of the evidence was assessed using the GRADE approach.
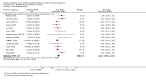
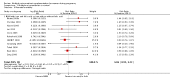
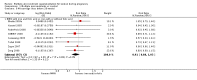
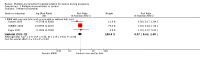
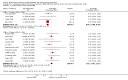
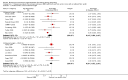
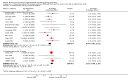
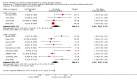
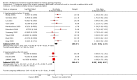
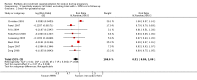
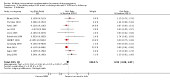
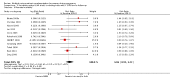
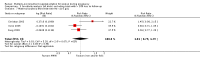
Main results: Nineteen trials (involving 138,538 women) were identified as eligible for inclusion in this review but only 17 trials (involving 137,791 women) contributed data to the review. Fifteen of these 17 trials were carried out in low and middle-income countries and compared MMN supplements with iron and folic acid versus iron with or without folic acid. Two trials carried out in the UK compared MMN with a placebo. MMN with iron and folic acid versus iron, with or without folic acid (15 trials): MMN resulted in a significant decrease in the number of newborn infants identified as low birthweight (LBW) (average risk ratio (RR) 0.88, 95% confidence interval (CI) 0.85 to 0.91; high-quality evidence) or small-for-gestational age (SGA) (average RR 0.92, 95% CI 0.86 to 0.98; moderate-quality evidence). No significant differences were shown for other maternal and pregnancy outcomes: preterm births (average RR 0.96, 95% CI 0.90 to 1.03; high-quality evidence), stillbirth (average RR 0.97, 95% CI 0.87, 1.09; high-quality evidence), maternal anaemia in the third trimester (average RR 1.03, 95% CI 0.85 to 1.24), miscarriage (average RR 0.91, 95% CI 0.80 to 1.03), maternal mortality (average RR 0.97, 95% CI 0.63 to 1.48), perinatal mortality (average RR 1.01, 95% CI 0.91 to 1.13; high-quality evidence), neonatal mortality (average RR 1.06, 95% CI 0.92 to 1.22; high-quality evidence), or risk of delivery via a caesarean section (average RR 1.04; 95% CI 0.74 to 1.46).A number of prespecified, clinically important outcomes could not be assessed due to insufficient or non-available data. Single trials reported results for: very preterm birth < 34 weeks, macrosomia, side-effects of supplements, nutritional status of children, and congenital anomalies including neural tube defects and neurodevelopmental outcome: Bayley Scales of Infant Development (BSID) scores. None of these trials reported pre-eclampsia, placental abruption, premature rupture of membranes, cost of supplementation, and maternal well-being or satisfaction.When assessed according to GRADE criteria, the quality of evidence for the review's primary outcomes overall was good. Pooled results for primary outcomes were based on multiple trials with large sample sizes and precise estimates. The following outcomes were graded to be as of high quality: preterm birth, LBW, perinatal mortality, stillbirth and neonatal mortality. The outcome of SGA was graded to be of moderate quality, with evidence downgraded by one for funnel plot asymmetry and potential publication bias.We carried out sensitivity analysis excluding trials with high levels of sample attrition (> 20%); results were consistent with the main analysis except for the findings for SGA (average RR 0.91, 95% CI 0.84 to 1.00). We explored heterogeneity through subgroup analyses by maternal height and body mass index (BMI), timing of supplementation and dose of iron. Subgroup differences were observed for maternal BMI for the outcome preterm birth, with significant findings among women with low BMI. Subgroup differences were also observed for maternal BMI and maternal height for the outcome SGA, indicating a significant impact among women with higher maternal BMI and height. The overall analysis of perinatal mortality, although showed a non-significant effect of MMN supplements versus iron with or without folic acid, was found to have substantial statistical heterogeneity. Subgroup differences were observed for timing of supplementation for this outcome, indicating a significantly higher impact with late initiation of supplementation. The findings between subgroups for other primary outcomes were inconclusive. MMN versus placebo (two trials): A single trial in the UK found no clear differences between groups for preterm birth, SGA, LBW or maternal anaemia in the third trimester. A second trial reported the number of women with pre-eclampsia; there was no evidence of a difference between groups. Other outcomes were not reported.
Authors' conclusions: Our findings support the effect of MMN supplements with iron and folic acid in improving some birth outcomes. Overall, pregnant women who received MMN supplementation had fewer low birthweight babies and small-for-gestational-age babies. The findings, consistently observed in several systematic evaluations of evidence, provide a basis to guide the replacement of iron and folic acid with MMN supplements containing iron and folic acid for pregnant women in low and middle-income countries where MMN deficiencies are common among women of reproductive age. Efforts could focus on the integration of this intervention in maternal nutrition and antenatal care programs in low and middle-income countries.
Conflict of interest statement
Batool A Haider: none.
Zulfiqar A Bhutta was the principal investigator of the UNIMAPP trial conducted in Pakistan (Bhutta 2009a). He was not involved in the screening and data extraction for this paper, which was conducted by other review authors acknowledged above.
Figures

























Update of
-
Multiple-micronutrient supplementation for women during pregnancy.Cochrane Database Syst Rev. 2015 Nov 1;2015(11):CD004905. doi: 10.1002/14651858.CD004905.pub4. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2017 Apr 13;4:CD004905. doi: 10.1002/14651858.CD004905.pub5. PMID: 26522344 Free PMC article. Updated. Review.
Similar articles
-
Multiple-micronutrient supplementation for women during pregnancy.Cochrane Database Syst Rev. 2015 Nov 1;2015(11):CD004905. doi: 10.1002/14651858.CD004905.pub4. Cochrane Database Syst Rev. 2015. Update in: Cochrane Database Syst Rev. 2017 Apr 13;4:CD004905. doi: 10.1002/14651858.CD004905.pub5. PMID: 26522344 Free PMC article. Updated. Review.
-
Multiple-micronutrient supplementation for women during pregnancy.Cochrane Database Syst Rev. 2019 Mar 14;3(3):CD004905. doi: 10.1002/14651858.CD004905.pub6. Cochrane Database Syst Rev. 2019. PMID: 30873598 Free PMC article.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Multiple-micronutrient supplementation for women during pregnancy.Cochrane Database Syst Rev. 2012 Nov 14;11:CD004905. doi: 10.1002/14651858.CD004905.pub3. Cochrane Database Syst Rev. 2012. Update in: Cochrane Database Syst Rev. 2015 Nov 01;(11):CD004905. doi: 10.1002/14651858.CD004905.pub4. PMID: 23152228 Updated. Review.
-
Effects of vitamin and mineral supplementation during pregnancy on maternal, birth, child health and development outcomes in low- and middle-income countries: A systematic review.Campbell Syst Rev. 2021 Jun 26;17(2):e1127. doi: 10.1002/cl2.1127. eCollection 2021 Jun. Campbell Syst Rev. 2021. PMID: 37051178 Free PMC article.
Cited by
-
Determinants of micronutrient supplementation during pregnancy among women in three sub-Saharan African countries: a multilevel logistic regression model.Front Glob Womens Health. 2024 Oct 25;5:1449259. doi: 10.3389/fgwh.2024.1449259. eCollection 2024. Front Glob Womens Health. 2024. PMID: 39524499 Free PMC article.
-
Associations of Maternal Nutritional Status and Supplementation with Fetal, Newborn, and Infant Outcomes in Low-Income and Middle-Income Settings: An Overview of Reviews.Nutrients. 2024 Oct 31;16(21):3725. doi: 10.3390/nu16213725. Nutrients. 2024. PMID: 39519557 Free PMC article. Review.
-
A formative study of the sociocultural influences on dietary behaviours during pregnancy in rural Bangladesh.Matern Child Nutr. 2024 Oct;20 Suppl 6(Suppl 6):e13713. doi: 10.1111/mcn.13713. Epub 2024 Aug 30. Matern Child Nutr. 2024. PMID: 39212336 Free PMC article.
-
Daily oral iron supplementation during pregnancy.Cochrane Database Syst Rev. 2024 Aug 15;8(8):CD004736. doi: 10.1002/14651858.CD004736.pub6. Cochrane Database Syst Rev. 2024. PMID: 39145520 Review.
-
Effect of the treatment of iron deficiency anemia on chronic drug-resistant cough: a rare case report.Daru. 2024 Dec;32(2):967-971. doi: 10.1007/s40199-024-00522-1. Epub 2024 Jun 8. Daru. 2024. PMID: 38850453
References
References to studies included in this review
-
- Bhutta ZA, Rizvi A, Raza F, Hotwani S, Zaidi S, Soofi S, et al. A comparative evaluation of multiple micronutrient and iron‐folate supplementation during pregnancy in Pakistan: impact on pregnancy outcomes. Food and Nutrition Bulletin 2009;30(4):S496‐S505. - PubMed
- Persson LA, Eneroth H, Ekstrom EC. Multiple micronutrient supplementation during pregnancy: a review of effects on birth size, maternal haemoglobin and perinatal mortality demonstrated in trials in Bangladesh, Guinea‐Bissau and Pakistan. Report for UNICEF/UNU/WHO2004.
-
- Brough L, Rees GA, Crawford MA, Morton RH, Dorman EK. Effect of multiple‐micronutrient supplementation on maternal nutrient status, infant birth weight and gestational age at birth in a low‐income, multi‐ethnic population. British Journal of Nutrition 2010;104(3):437‐45. - PubMed
-
- Christian P, Darmstadt GL, Wu L, Khatry SK, LeClerq SC, Katz J, et al. The effect of maternal micronutrient supplementation on early neonatal morbidity in rural Nepal: a randomised, controlled, community trial. Archives of Disease in Childhood 2008;93(8):660‐4. - PubMed
- Christian P, Jiang T, Khatry SK, LeClerq SC, Shrestha SR, West KP Jr. Antenatal supplementation with micronutrients and biochemical indicators of status and subclinical infection in rural Nepal. American Journal of Clinical Nutrition 2006;83:788‐94. - PubMed
- Christian P, Khatry SK, Katz J, Pradhan EK, LeClerq SC, Shrestha SR, et al. Effects of alternative maternal micronutrient supplements on low birth weight in rural Nepal: double blind randomised community trial. BMJ 2003;326:571. - PMC - PubMed
- Christian P, Khatry SK, LeClerq SC, Dali SM. Effects of prenatal micronutrient supplementation on complications of labor and delivery and puerperal morbidity in rural Nepal. International Journal of Gynaecology and Obstetrics 2009;106(1):3‐7. - PubMed
- Christian P, Murray‐Kolb LE, Khatry SK, Katz J, Schaefer BA, Cole PM, et al. Prenatal micronutrient supplementation and intellectual and motor function in early school‐aged children in Nepal. JAMA 2010;304(24):2716‐23. - PubMed
- Christian P, Nanayakkara‐Bind A, Schulze K, Wu L, LeClerq SC, Khatry SK. Antenatal micronutrient supplementation and third trimester cortisol and erythropoietin concentrations. Maternal & Child Nutrition 2014 [Epub ahead of print]. - PMC - PubMed
- Christian P, Shrestha J, LeClerq SC, Khatry SK, Jiang T, Wagner T, et al. Supplementation with micronutrients in addition to iron and folic acid does not further improve the hematologic status of pregnant women in rural Nepal. Journal of Nutrition 2003;133:3492‐8. - PubMed
- Christian P, Stewart CP, LeClerq SC, Wu L, Katz J, West KP Jr, et al. Antenatal and postnatal iron supplementation and childhood mortality in rural Nepal: a prospective follow‐up in a randomized, controlled community trial. American Journal of Epidemiology 2009;170(9):1127‐36. - PMC - PubMed
- Christian P, West KP Jr, Khatry SK, LeClerq SC, Pradhan EK, Katz J, et al. Effects of maternal micronutrient supplementation on fetal loss and infant mortality: a cluster‐randomised trial in Nepal. American Journal of Clinical Nutrition 2003;78:1194‐202. - PubMed
- Katz J, Christian P, Dominici F, Zeger SL. Treatment effects of maternal micronutrient supplementation vary by percentiles of the birth weight distribution in rural Nepal. Journal of Nutrition 2006;136:1389‐94. - PubMed
- Kulkarni B, Christian P, LeClerq SC, Khatry SK. Determinants of compliance to antenatal micronutrient supplementation and women's perceptions of supplement use in rural Nepal. Public Health Nutrition 2010;13(1):82‐90. - PubMed
- Murray‐Kolb L, Bahnfleth C, Hurley K, Cole P, Khatry S, LeClerq S, et al. Micronutrient supplementation during pregnancy and the preschool years and child behavior at 7‐9 years of age. FASEB Journal 2014;28(1 Suppl):Abstract no: 619.3.
- Nanayakkara‐Bind A, Schulze K, Wu L, Le SC, Khatry SK, Christian P. Effects of antenatal micronutrient supplementation on cortisol and erythropoietin in pregnant Nepalese women. FASEB Journal 2011;25:Abstract no: 779.15.
- Stewart CP, Christian P, LeClerq SC, West KP Jr, Khatry SK. Antenatal supplementation with folic acid + iron + zinc improves linear growth and reduces peripheral adiposity in school‐age children in rural Nepal. American Journal of Clinical Nutrition 2009;90(1):132‐40. - PMC - PubMed
- Stewart CP, Christian P, Schulze KJ, Arguello M, Leclerq SC, Khatry SK, et al. Low maternal vitamin B‐12 status is associated with offspring insulin resistance regardless of antenatal micronutrient supplementation in rural Nepal. Journal of Nutrition 2011;141(10):1912‐7. - PubMed
- Stewart CP, Christian P, Schulze KJ, Leclerq SC, West KP Jr, Khatry SK. Antenatal micronutrient supplementation reduces metabolic syndrome in 6‐ to 8‐year‐old children in rural Nepal. Journal of Nutrition 2009;139(8):1575‐81. - PubMed
-
- Fawzi WW, Msamanga GI, Urassa W, Hertzmark E, Petraro P, Willett WC, et al. Vitamins and perinatal outcomes among HIV‐negative women in Tanzania. New England Journal of Medicine 2007;356(14):1423‐31. - PubMed
-
- Friis H, Gomo E, Nyazema N, Ndhlovu P, Krarup H, Kaestel P, et al. Effect of micronutrient supplementation on gestational length and birth size: a randomized, placebo‐controlled, double‐blind effectiveness trial in Zimbabwe. American Journal of Clinical Nutrition 2004;80:178‐84. - PubMed
References to studies excluded from this review
-
- Agarwal N, Dora S, Kriplani A, Garg P, Vivekanandhan S, Kulshrestha V. Response of therapy with vitamin B6, B12 and folic acid on homocysteine level and pregnancy outcome in hyperhomocysteinemia with unexplained recurrent abortions. International Journal of Gynecology and Obstetrics 2012;119(Suppl 3):S759.
-
- Aguayo VM, Kone D, Bamba SI, Diallo B, Sidibe Y, Traore D, et al. Acceptability of multiple micronutrient supplements by pregnant and lactating women in Mali. Public Health Nutrition 2005;8(1):33‐7. - PubMed
-
- Ahn E, Pairaudeau N, Pairaudeau N, Cerat Y, Couturier B, Fortier A, et al. A randomized cross over trial of tolerability and compliance of a micronutrient supplement with low iron separated from calcium vs high iron combined with calcium in pregnant women. BMC Pregnancy and Childbirth 2006;6:10. - PMC - PubMed
-
- An H, Yin S, Xu Q. Effects of supplementing calcium, iron and zinc on the fetus development and growth during pregnancy. Chinese Journal of Preventive Medicine 2001;35(6):370‐3. - PubMed
References to studies awaiting assessment
-
- Adu‐Afarwuah S, Lartey A, Okronipa H, Maleta K, Ashorn P, Ashorn U, et al. Efficacy of LNS products for pregnant and lactating women: pregnancy outcomes from the iLiNS‐dyad studies in Ghana and Malawi. Annals of Nutrition and Metabolism 2013;63:24‐5.
- Ashorn P, Alho L, Ashorn U, Cheung YB, Dewey KG, Harjunmaa U, et al. The impact of lipid‐based nutrient supplement provision to pregnant women on newborn size in rural Malawi: a randomized controlled trial. American Journal of Clinical Nutrition 2015;101(2):387‐97. - PubMed
-
- Gathwala G. Effect of antenatal multiple micronutrient supplementation of mothers on the birthweight of their infants. Journal of Paediatrics and Child Health 2012;48(Suppl 1):47‐8.
References to ongoing studies
-
- Biggs BA. A randomised controlled trial to compare the impact on birth weight of daily iron‐folic acid, twice weekly iron‐folic acid and twice weekly multiple micronutrient supplementation for pregnant women in Ha Nam province, Vietnam. Australian New Zealand Clinical Trials Registry (www.anzctr.org.au) (acc...2011.
-
- Adu‐Afarwuah S, Lartey A, Okronipa H, Maleta K, Ashorn P, Ashorn U, et al. Efficacy of LNS products for pregnant and lactating women: Pregnancy outcomes from the iLiNS‐dyad studies in Ghana and Malawi. Annals of Nutrition and Metabolism 2013;63:24‐5.
- Adu‐Afarwuah S, Lartey A, Okronipa H, Peerson J, Vosti S, Ashorn P, et al. Lipid‐based nutrient supplement for pregnant women improve birth outcomes among primiparous but not multiparous women in Ghana. FASEB Journal 2014;28(1 Suppl 1):Abstract no: 256.7.
- Dewey KG. Efficacy of lipid‐based nutrient supplements (LNS) for pregnant and lactating women and their infants. ClinicalTrials.gov (http://clinicaltrials.gov/) (accessed 15 February 2011)2011. - PMC - PubMed
- Oaks B, Adu‐Afarwuah S, Lartey A, Stewart C, Ashorn P, Vosti S, et al. Lipid‐based nutrient supplementation during pregnancy decreases maternal cortisol in younger women. FASEB Journal 2014;28(1 Suppl 1):Abstract no. 389.6.
-
- Hirschberg AL. Supplementation with WellnessPack mama during pregnancy and lactation‐ a randomized double‐blind, placebo‐controlled study. ClinicalTrials.gov (http://clinicaltrials.gov/) [accessed 2 September 2014]2014.
-
- Jobarteh ML, Moore S, Kennedy C, Gambling L, McArdle HJ. The effect of nutritional supplementation during pregnancy on placental transporter expression in placentas from gambian women. Placenta 2013;34(9):A81‐2.
- Moore S. Investigating the effects of pre‐natal and infancy nutritional supplementation on infant immune development in The Gambia: the Early Nutrition and Immune Development (ENID) trial. Current Clinical Trials (http://www.current‐trials.com) (accessed 8 July 2011). - PMC - PubMed
- Moore SE, Fulford AJC, Darboe MK, Jobarteh ML, Jarjou LM, Prentice AM. A randomized trial to investigate the effects of pre‐natal and infant nutritional supplementation on infant immune development in rural Gambia: The ENID trial: Early Nutrition and Immune Development. BMC Pregnancy and Childbirth 2012;12:107. - PMC - PubMed
-
- Harding KL, Matias SL, Moniruzzaman M, Stewart CP, Mridha MK, Vosti SA, et al. Rang‐Din Nutrition Study: assessment of participant adherence to lipid‐based nutrient and iron‐folic acid supplements among pregnant and lactating women in the context of a study on the effectiveness of supplements in Bangladesh. FHI 360/FANTA. Washington, DC: US Agency for International Development (USAID), 2014.
- Mridha M, Chaparro C, Paul R, Hussain S, Vosti S, Matias S, et al. Lipid‐based nutrient supplements for pregnant women reduce newborn stunting in Bangladesh. FASEB Journal 2014;28(1 Suppl):Abstract no: 256.6. - PMC - PubMed
Additional references
-
- Allen LH. Multiple micronutrients in pregnancy and lactation: an overview. American Journal of Clinical Nutrition 2005;81(5):1206S–1212S. - PubMed
-
- WHO Secretariat, Andersson M, Benoist B, Delange F, Zupan J. Prevention and control of iodine deficiency in pregnant and lactating women and in children less than 2‐years‐old: conclusions and recommendations of the technical consultation. Public Health Nutrition 2007;10(12A):1606‐11. - PubMed
-
- Benoist B, McLean E, Andersson M, Rogers L. Iodine deficiency in 2007: global progress since 2003. Food and Nutrition Bulletin 2008;29(3):195‐202. - PubMed
-
- Berti C, Biesalski HK, Gärtner R, Lapillonne A, Pietrzik K, Poston L, et al. Micronutrients in pregnancy: current knowledge and unresolved questions. Clinical Nutrition (Edinburgh, Scotland) 2011;30(6):689‐701. - PubMed
-
- Bhutta ZA, Haider BA. Maternal micronutrient deficiencies in developing countries. Lancet 2008;371(9608):186‐7. - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical


