Highlights
-
•
Corona virus infections can cause Acute Respiratory Distress Syndrome (ARDS), mediated by a variety of mechanisms, including cytokine storm, dysregulation of the renin-angiotensin system, neutrophil activation, and the coagulation cascade.
-
•
Vitamin D receptor activation act on many cellular and biochemical mechanisms responsible for ARDS. Most data suggest that VDR activation may taper down ARDS.
-
•
Vitamin D supplementation, either as vitamin D or calcifediol (25OHD), is being tested in several RCTs as to demonstrate its potential effects on the course of corona virus infections.
-
•
A pilot study in Cordoba suggested that calcifediol treatment may reduce the need for ICU treatment of patients hospitalized for COVID19 infections.
Keywords: Vitamin D, vitamin D3 or cholecalciferol; Calcifediol or 25-hydroxyvitamin D3; 1α, 25(OH)2D or 1α, 25-dihydroxyvitamin D or calcitriol; Corona virus; SARS-CoV-2; Acute respiratory distress syndrome (ARDS); Cytokine storm; Lung diseases; Renin-angiotensin system; Hypercoagulability
Abstract
Coronavirus infection is a serious health problem awaiting an effective vaccine and/or antiviral treatment. The major complication of coronavirus disease 2019 (COVID-19), the Acute Respiratory Distress syndrome (ARDS), is due to a variety of mechanisms including cytokine storm, dysregulation of the renin-angiotensin system, neutrophil activation and increased (micro)coagulation. Based on many preclinical studies and observational data in humans, ARDS may be aggravated by vitamin D deficiency and tapered down by activation of the vitamin D receptor. Several randomized clinical trials using either oral vitamin D or oral Calcifediol (25OHD) are ongoing. Based on a pilot study, oral calcifediol may be the most promising approach. These studies are expected to provide guidelines within a few months.
1. Introduction
The coronavirus disease 2019 (COVID-19) is rapidly causing worldwide morbidity and mortality. While most infected people will recover after a mild to modest course of the disease, some patients, especially older people or those with other major diseases, will suffer serious morbidity and a high mortality risk. In the absence of vaccines, some therapeutic interventions have some proven benefits (such as convalescent plasma [1] and remdesivir [2], whereas other approaches (including vitamin D) that may influence the course of the disease deserve special attention.
The vitamin D endocrine system is well known for its beneficial effects on calcium and bone homeostasis, especially in children and elderly subjects. Moreover, it may have several extra-skeletal effects [3] especially on the immune system and lung function. All cells of the immune cells can express the vitamin D receptor (VDR) and most cytokines, produced by or regulating these immune cells are under the coherent control of the active vitamin D hormone, 125(OH)2D. Indeed, in essence, 1α,25(OH)2D activates the native immune defense system while tapering down the acquired immune system [[3], [4], [5]]. In addition, antigen-presenting cells and monocytic cells can express CYP27B1, the essential enzyme for the local, auto/paracrine production of 1α,25(OH)2D in the immune system. Vitamin D deficiency may predispose to increased risk of infections, and vitamin D supplementation may decrease the risk of upper respiratory infections [6]. The lung epithelium also expresses the VDR and CYP27B1 and may be an important target tissue for the vitamin D endocrine system [7]. Therefore, there may be many potential links between viral infections such as COVID19 and vitamin D status.
In this Viewpoint, we summarize how activation of the vitamin D Receptor (VDR) may be able to decrease acute lung injury (ALI) and Acute Respiratory Distress Syndrome (ARDS). We therefore will first review the major mechanisms underlying ALI and ARDS in patients with viral (including Coronavirus) infections. Thereafter, we present an overview of the ongoing RCTs that hopefully will allow to better describe the implications of vitamin D deficiency or its rapid correction for the course of this disease.
2. Major mechanism involved in the pathogenesis of ARDS
The occurrence and severity of Acute Respiratory Distress Syndrome (ARDS) in patients with Coronavirus Disease 2019 (COVID‐19) is a life‐threatening condition and a major determining factor of the prognosis [8]. Patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)–associated pneumonitis or ARDS have pulmonary inflammation, thick mucus secretions in the airways, elevated levels of proinflammatory cytokines, extensive lung damage, and microthrombosis [9]. Almost 20 % of the hospitalized patients (including ICU and non‐ICU patients) with COVID‐19 developed ARDS and, despite recent improvements in mechanical ventilation strategies and supportive care, about 65 % of patients with ARDS died [10].In ARDS, the epidemiological and morbidity/mortality patterns are similar regardless of the trigger [11]. ARDS is a pivotal component of the pathophysiological processes by which patients with severe COVID‐19 proceed to develop multiple organ dysfunction with high mortality [8]. ARDS onset is often rapid and progressive, it appears approximately nine days after the onset of severe COVID‐19 infection [12], and patients with ARDS died a mean of 20 days after the onset of the symptoms or about 9–11 days after ICU admission [13]. The disease is difficult to manage at a late stage of the disease and early treatment is thus critical to control the progression of infection and improving the prognosis of patients with ARDS. There is currently no Food and Drug Administration-approved effective pharmacologic treatment for ARDS, and management remains supportive with lung-protective mechanical ventilation [14,15]. ARDS is the common immune-pathological event for SARS-CoV-2, SARS-CoV, and MERS-CoV infections [8]. The main pathophysiological mechanisms involved in ARDS are:
-
1)
A cytokine storm or the uncontrolled, sometimes fatal, inflammatory response resulting from the release of large amounts of pro-inflammatory cytokines (IFN-α, IFN-γ, IL-1β, IL-6, IL-12, IL-18, IL-33, TNF-α, TGFβ, etc.) and chemokines (CCL2, CCL3, CCL5, CXCL8, CXCL9, CXCL10, etc.) by immune effector cells during SARS-CoV infection [16,17]. Similar to those infected with SARS-CoV, patients with severe MERS-CoV infection showed elevated levels of serum IL-6, IFN-α, and CCL5, CXCL8, CXCL-10 compared to those with mild-moderate disease [18]. In addition, the excessive activation and recruitment of neutrophils into inflamed interstitium and alveolar space with disruption of the endothelial-epithelial barrier and alveolar damage exacerbates the pathogenesis of ARDS and may indicate a poor clinical outcome [19]. The chemokine (C-X-C motif) ligand 8 (CXCL8) and interleukin-8, the chemokine that modulates innate and adaptive immune responses by recruiting inflammatory cells (i.e., neutrophils, T lymphocytes and NK cells) to the sites of inflammation, are considered to be the archetypal neutrophil chemoattractants [20]. By binding to its receptor CXCR3, CXCL10 can induce chemotaxis, apoptosis, cell growth and angiostasis [21]. The limited data so far in patients infected with SARS-CoV-2 confirmed the presence of high amounts of pro-inflammatory cytokines and chemokines. Moreover, patients requiring ICU admission had higher concentrations of GCSF, IP10, MCP1, MIP1A, and TNFα than did those not requiring ICU admission, suggesting that the cytokine storm was associated with disease severity [9].
-
2)
The activation of the renin angiotensin system (RAS) with decrease of angiotensin converting enzyme ACE2 have been implicated in the pathogenesis of acute respiratory distress syndrome [22]. Indeed, local or systemic inflammatory reactions may activate the RAS whereby angiotensin II generated by ACE is able to induce lung damage, whereas ACE2 transforms angiotensin II into smaller peptides with lung protective effects. The alteration of the balance between the levels of the enzymes ACE and ACE2 that affects the endogenous ratio of Ang II: Ang-(1–7), with elevation of angiotensin II, is key to the development of ALI and ARDS in both animal models [22,23] and humans [24,25]. It is important to highlight that SARS-CoV-2 does not use other coronavirus receptors such as aminopeptidase N and dipeptidyl peptidase 4 [26], but SARS-CoV-2 uses ACE2 as a cellular entry receptor [27]. 83 % of cells expressing ACE2 are type II alveolar epithelial cells (AECII), suggesting that these cells may serve as a reservoir for viral invasion [28]. Interestingly, Kuba et al [26] found that the expression of ACE2 (but not the ACE) in lung tissues was significantly downregulated in mice, decreasing the availability of ACE2 during SARS-CoV-2-induced lung injury. ACE2 counter-regulates the effects produced by Ang II by converting Ang II to Ang-(1–7), activates Mas to repress the signaling pathways of STAT3 and extracellular signal-regulated kinases (ERK) [29], and acts as an anti-inflammatory factor [30]. In this way, a lung protective mechanism is lost and SARS-CoV infection becomes more lethal, with exaggerated neutrophil accumulation, increased pulmonary vascular permeability and exacerbated pulmonary edema, which eventually lead to ARDS. Both mechanisms lead to an extensive inflammatory process, with activation, recruitment and influx of activated neutrophils and macrophages in the alveolar space due to the disruption of the alveolar epithelial barrier with diffuse alveolar damage. The alveolus is filled with activated neutrophils, cytokines/chemokines and protein-rich exudate. In this state, the lungs cannot provide enough oxygen to the blood for the body's vital organs [8]. Moreover, the state of hyper-inflammation and activation of the RAS are intimately involved in altering the coagulation cascade [31], which in cooperation with endothelial cell infection and endotheliitis [32] leads to a more prothrombotic status seen in ARDS during SARS-CoV-2 infections. The dysregulation of the coagulation cascade and the subsequent formation of intra-alveolar or systemic fibrin clots and thrombotic complications are prominent findings in coronavirus infections associated with ARDS [33,34]. Another consequence is the potential risk of fibrosis [35] with impaired lung function after recovery in the course of natural history of the disease [36].
3. Potential role of activation of the VDR in ARDS
Vitamin D deficiency is associated with an increased risk of upper respiratory infection. A recent meta-analysis using about 10,000 individual participants data from 25 RCTs, concluded that vitamin D supplementation reduced the risk of upper respiratory infections by about 19 % [6]. Patients with severe vitamin D deficiency experienced the greatest benefit. Whether this conclusion would also apply to corona virus infections is unknown and will not be discussed here as we want to focus on the potential role of vitamin D on ARDS.
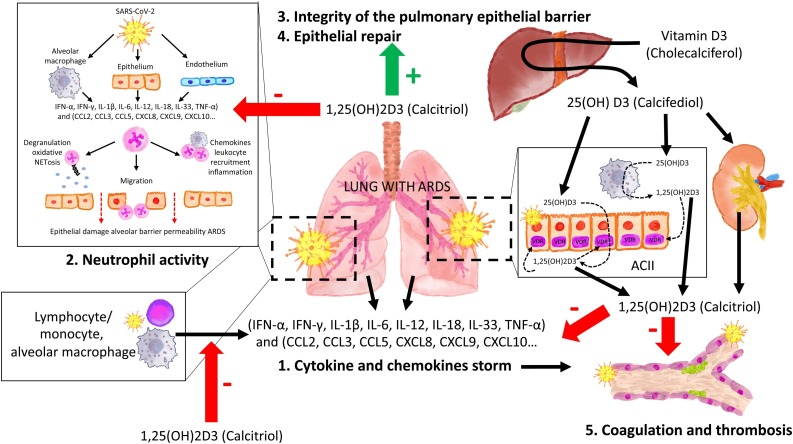
Several studies have suggested that the vitamin D/VDR signaling pathway may provide some beneficial effects in LPS-induced ARDS mediated by several mechanism (Fig. 1, Fig. 2 ) such as (1) decreasing the storm of cytokines and chemokines; (2) regulating the renin‑angiotensin system; (3) modulating neutrophil activity; (4) maintaining the integrity of the pulmonary epithelial barrier and (5) stimulating epithelial repair [[37], [38], [39], [40]].
Fig. 1.
The mechanisms involved in the pathogenesis of Acute Respiratory Distress Syndrome (ARDS) including cytokine and chemokine storm (release of large amounts by immune effector cells), Excessive activation and recruitment of neutrophils into inflamed interstitium and alveolar space with disruption of the endothelial-epithelial barrier and alveolar damage, and dysregulation of the coagulation cascade generating intra-alveolar or systemic fibrin clots and thrombotic complications.
The vitamin D endocrine system minimizes ARDS. The vitamin D receptor (VDR) and enzymes of the vitamin D endocrine system are expressed in the cuboidal alveolar type II cells (ACII) and monocyte/macrophages and activated lymphocytes. The availability of calcifediol is critical for synthesizing calcitriol, which through endocrine, auto/paracrine action on VDR: 1) decreases the intensity of Cytokine and Chemokine storm, 2)modulating neutrophil activity, 3)maintaining the integrity of the pulmonary epithelial barrier, 4)stimulating epithelial repair and 5)decreasing directly and indirectly the risk of hypercoagulability and pulmonary or systemic thrombosis
SARS-CoV-2: severe acute respiratory syndrome coronavirus 2. IFN-α, IFN-γ: Interferon gamma α and γ; IL-1β, IL-6, IL-12, IL-18, IL-33 (Interleukin -1β, 6, 12, 18, 33) TNF-α (Tumor Necrosis Factor-α). TGFβ (Transforming growth factor α and β). CCL2, CCL3, CCL5 Chemokine (C-C motif) ligand 2,3.5) CXCL8, CXCL9, CXCL10: C-X-C (motif chemokine ligand 8,910).
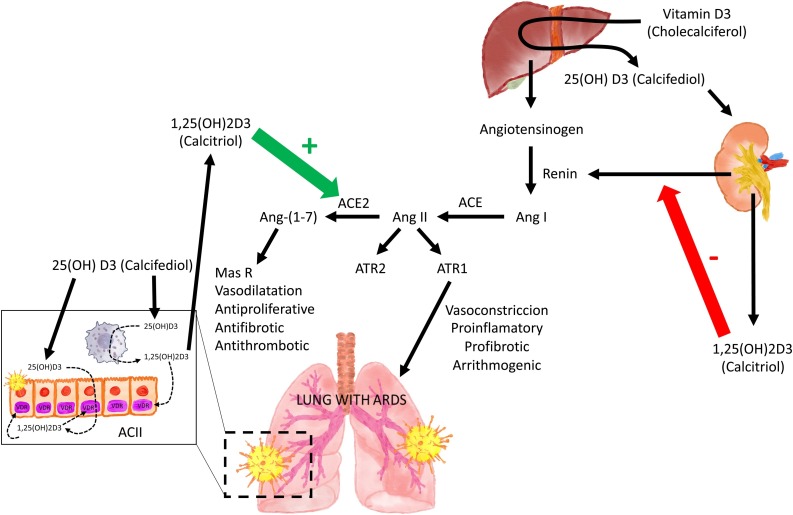
Fig. 2.
The Renin Angiotensin System (RAS) and Acute Respiratory Distress Syndrome (ARDS).
Local or systemic inflammatory reactions may activate RAS and ACE thereby generating angiotensin II, which via its receptor (ATR) is able to induce lung damage. During SARS-CoV-2 invasion ACE2 is downregulated in Type II alveolar epithelial cells thereby decreasing the conversion of Ang II to Ang-(1–7). This prevents the protective action of the Ang (1–7), acting on its receptor (Mas R), and all aspects of ARDS.
1α,25(OH)2D/VDR is a powerful negative regulator of the renin-angiotensin system (RAS) inhibiting renin and the ACE/Ang II/AT1R cascade and inducing ACE2/Ang-(1–7) axis activity.
ACII: cuboidal alveolar type II cells. SARS-CoV-2: severe acute respiratory syndrome coronavirus 2 Ang I angiotensin I. Ang II: angiotensin II. Ang-(1–7) angiotensin1–7. MasR: G protein-coupled Mas receptor. AT1R y AT2R: angiotensin II receptor 1 and 2.
(1) VDR is highly expressed in the cuboidal alveolar type II cells (ACII) of the lung [7] [37]. Overexpression of VDR exerts anti-inflammatory effects in the lung [41]. VDR-knockout mice experienced more severe acute lung injury (ALI) than wild-type mice, following LPS treatment. The endocrine system of vitamin D has been shown, in various in vitro models, to inhibit the production and release of cytokines (IFN-α, IFN-γ, IL-1β, IL-6, IL-12, IL-18, IL-33, TNF-α) and chemokines (CCL2, CCL3, CCL5, CXCL8, CXCL9, CXCL10 involving ARDS [7,[42], [43], [44], [45], [46]]. The calcitriol/VDR signaling may also protect against ALI by inhibiting the angiopoietin-2-TEK receptor tyrosine kinase-myosin light-chain kinase pathway [39]. Thus, 1α,25(OH)2D is important in maintaining the structure and function of epithelial barriers in multiple tissues [47] mediated by alveolar epithelial tight junctions and gene regulation of occludin and zonula occludens-1 (ZO-1) expression [38]. 1α,25(OH)2D also inhibits neutrophil recruitment in an animal model of acute lung injury, due to its inhibitory effect on cytokines [48].
(2) There is ample evidence that 1α,25(OH)2D/VDR is a powerful negative regulator of renin-angiotensin system (RAS). Indeed, renin is increased in VDR null mice [49]. Similarly, 1α-hydroxylase-deficient mice exhibit increased activity of the intrarenal RAS that is downregulated with the administration of 1α,25(OH)2D [38]. Chronic vitamin D deficiency may induce RAS activation [50]. 1α,25(OH)2D inhibits renin, ACE and Ang II expression, and induces ACE2 levels in LPS-induced ALI. In addition, dysregulation of local and circulating RAS, with enhanced ACE/Ang II expression levels and reduced ACE2/Ang-(1–7) expression levels, was reported to contribute to ischemia-reperfusion-induced ALI in mice [51]. Therefore, vitamin D may attenuate LPS-Induced ALI by, at least partially, inducing ACE2/Ang-(1–7) axis activity and inhibiting renin and the ACE/Ang II/AT1R cascade (Fig. 2) [37]. VDR activation is also able to inhibit the protein Skp2 [52,53] which plays a central role in the mechanism of viral replication of the COVID-19. Indeed, COVID-19 uses blockade of autophagy for accelerated replication and infectivity [54]. To achieve this, the virus induces Skp2, which, in turn, inactivates Beclin 1, an essential component of the autophagic process. 1α,25(OH)2D also stimulate the production of Klotho, known to attenuate multiorgan aging and increase longevity also promotes autophagy through the maintenance of adequate cellular levels of Beclin [55].
A caveat about the potential beneficial effects of VDR activation on the RAS system is, however, needed as SARS-CoV-2 spike protein binds with high affinity to ACE2 from humans, ferrets, cats and other species and use this membrane protein as cellular entry mechanism [56]. As 1α,25(OH)2D upregulates ACE2 it may thus facilitate the uptake of the virus in cells expressing this enzyme on their membranes. This dual effect of ACE2 being potentially harmful or beneficial for virus induced lung (or other tissue) damage has generated intensive debate also related to the use of angiotensin antagonists in hypertensive patients [57]. Overall, most experts concluded that the potential beneficial effects of ACE2 on heart and lungs may overrule its role in virus entrance [[58], [59], [60]].
4. Neutrophils, pneumonitis and vitamin D
Increased neutrophil infiltration is part of this picture of viral pneumonitis and ARDS and aims to destroy the infiltrating microorganisms by oxidative burst and phagocytosis. Neutrophils can also kill pathogens by the formation of Neutrophil Extracellular Traps (NETs) [61]. NETs are web-like structures of DNA and proteins expelled from the neutrophil that ensnare pathogens. Prior reports have linked aberrant NET formation to pulmonary diseases, thrombosis, mucous secretions in the airways and cytokine production [62]. At autopsy of a patient who died from corona virus pneumonia, intensive neutrophil infiltration and excess NETs were observed. Whether this is part of a defense strategy or whether suppression of such NET formation would be beneficial is unknown. The role of VDR activation in neutrophils has not been extensively studied. One study however reported that 125(OH)2D (in vitro) increased NET formation in neutrophils from normal subjects [63].
5. Maintaining the integrity of the pulmonary epithelial barrier and stimulating epithelial repair [[37], [38], [39], [40]]
The epithelial, cuboidal alveolar coating type II cells (ACII), which facilitate the entry of virus into the body, have high levels of multiple viral process-related genes, including regulatory genes for viral processes, viral life cycle, viral assembly, and viral genome replication [28]. Therefore, these cells are the major target cells for viral attack. As a result of the high basal expression of 1α-hydroxylase activation and low expression of the inactivating enzyme (24-hydroxylase), the ACIIs are able to convert circulating 25OHD into 1α,25(OH)2D [7], just as several cells of the immune system (antigen presenting cells and monocytic cells). 1α,25(OH)2D generated by ACII can act in an auto or paracrine fashion and, like cells of the immune system, increase the expression of vitamin D regulated genes with important innate immune functions (antimicrobial cathelicidin peptide gene and the TLR co-receptor CD14). In addition, in a viral infection model, dsRNA increased the expression of the 1α-hydroxylase and synergizes with calcifediol and calcitriol sequentially to induce cathelicidin [64]. Apart from defensins (such as cathelicidin), 1α,25(OH)2D also stimulates several genes in these ACII cells with favorable effects such as stimulation of surfactant factor and tight junction genes [65]. In a mouse model, 1α,25(OH)2D attenuated LPS-induced lung injury by promoting epithelial cell proliferation, and inhibited apoptosis and epithelial mesenchymal transition, suggesting that VDR activation may have therapeutic potential for the resolution of ARDS [40]. Similar beneficial effects were observed in other lung injury models [37,66].
6. Coagulation and thrombosis
Inflammation is an extremely complex pathophysiological process that is closely related to hemostasis. The activation of both systems is interdependent in a cycle of positive feedback, with one process promoting the effects of the other process and vice versa. Inflammation promotes a prothrombotic state. This crosstalk is mediated by pro-coagulant factors, pro-inflammatory cytokines, chemokines, adhesion molecules, tissue factor (TF) expression, platelets and endothelial cells [67]. The anti-thrombotic effects of vitamin D is well documented in preclinical studies. VDR knock out mice display increased platelet aggregation. Their gene expression of antithrombin (liver) and trombomodulin (aorta, liver and kidney) were downregulated, whereas tissue factor expression in liver and kidney were upregulated. VDR stimulation with 1α,25(OH)2D and/or its agonist maxacalcitol downregulated TF and upregulated thrombomodulin gene expression in monocytic cells, previously stimulated by tumor necrosis factor (TNF), lipopolysaccharides (LPS), and oxidized LDL (ox-LDL) [68]. VDRKO mice manifested exacerbated multi-organ thrombus formation after exogenous lipopolysaccharide injection with an increase in endothelial adhesion molecules, a decrease in NO production, and increased platelet aggregation [69]. 1α,25(OH)2D and its analogue paricalcitol significantly blunted the expression of TF, and its procoagulant activity, induced by the proinflammatory cytokine TNF-α in human aortic vascular smooth muscle cell (VSMCs), in a NF-κB-dependent manner. This was accompanied by the up-regulation of TF signaling mediator protease-activated receptor 2 (PAR-2) [70]. Observational data in humans revealed an association between low levels of 25OHD and the development of deep venous thromboembolic (DVT) [71] events in patients with ischemic stroke. Conversely, a significant positive association was found between serum levels 25OHD (> 20 ng/mL) and TF pathway inhibitor (TFPI) [a dual inhibitor of coagulation by binding to both TF/Factor VIIa complex as well as Factor Xa] [72].Most intervention studies however did not show clear benefits of vitamin D supplementation on major cardiovascular events, although most study subjects were not severely vitamin D deficient at baseline [3].
7. Clinical consequences: Vitamin D status and ADRS during corona virus infection
Despite the potential effects of VDR activation on ARDS or lung injury as described above, serious caution is needed before concluding that vitamin D supplementation may improve the outcome of SARS-CoV-2 infections. Indeed, as VDR/1α,25(OH)2D activation regulates a very large number of genes, mostly in cluster patterns, there has been a lot of speculations on the beneficial effects of vitamin D supplementation for a wide variety of (major) diseases, such as cancer, infections or diabetes [3,73]. However, several recent megatrials of vitamin D supplementation did not confirm such extra-skeletal health effects. The study participants, however, were mostly vitamin D replete adults and therefore it may well be that only (severely) vitamin D deficient subjects may benefit from vitamin D supplementation. There was one remarkable observation relevant to lung diseases. In a sub study of the New Zealand ViDA trial [74], vitamin D supplementation improved the lung function (expiratory volume in 1 s) in patients with asthma, COPD or in ever-smokers, especially when vitamin D deficient at baseline.
Vitamin D deficiency is highly prevalent around the world as about 7 % of the world population has severe deficiency and about 40 % live with modest deficiency [75]. In addition, patients with severe acute disease or respiratory distress syndrome (ARDS) [[76], [77], [78]] are even more deficient than control subjects. Studies are currently being carried out specifically on COVID-19 and vitamin D status [79,80]. Correction of vitamin D deficiency is relatively easy by either increased exposure to sunlight (unlikely for patients with ARDS), oral or parental vitamin D supplementation, or supplementation with 25OHD (calcifediol). To correct vitamin D deficiency in severely sick patients much higher doses than usual are needed [81], probably related to impaired hepatic conversion of vitamin D into 25OHD [82]. Calcifediol may have some advantages over the native vitamin D: it has a more reliable intestinal absorption (close to 100 %) and can rapidly restore serum concentrations of 25OHD as it does not require hepatic 25-hydroxylation. This is especially relevant in clinical situations whereby rapid restoration of serum 25OHD is desirable and CYP2R1 expression is compromised. Such impaired CYP2R1 activity has been well demonstrated in several animal models of obesity, diabetes or glucocorticoid excess [82]. This has also been demonstrated in patients with COPD or asthma [83]. In addition, calcifediol is about 3-fold more potent when compared to oral vitamin D3 in postmenopausal women [84]. An additional advantage of oral calcifediol is a more linear response curve, whereas there is a plateau effect with increasing doses of oral vitamin D3 [84]. The tissue effects of restoring vitamin D status may be due to circulating serum 125(OH)2D or, more likely, on the local conversion of 25OHD into the active hormone in pulmonary alveolar cells, immune cells or other potential target tissues.
8. Ongoing randomized controlled trials (RCTs)
It seems logical to correct vitamin D deficiency in all subjects for reasons unrelated to viral infection [6]. The available evidence suggests that the stimulation of VDR in patients with Coronavirus SARS-CoV-2 infection, may reduce Acute Respiratory Distress Syndrome (ARDS), with possibly beneficial effects on admission to intensive care unit (ICU) and deaths in the course of the disease. SARS-CoV-2 infection has been a challenge for clinicians involved in the diagnostic and therapeutic management of infected patients. The absence of specific treatment generated many trials but so far without final conclusions, except for some beneficial effect from treatment with convalescent plasma [1] or remdesivir [2,85]). The same holds true for testing the potential benefits of vitamin D or calcifediol supplementation of patients with SARS-CoV-2 infections. According to the NIH Trialnet database several observational and intervention studies are running:
1) Vitamin D on Prevention and Treatment of COVID-19 (NCT04334005) is a randomized, double-blind trial with a start date of April 10th 2020 and an end date of June 30th, 2020. The number of participants is 200 and the study population are patients infected with COVID-19. The intervention group will receive a single dose of 25,000 UI of vitamin D and the primary outcome measures are a composite of cumulative death (i.e. mortality) for all causes and for specific causes.
2) Low-risk, Early Aspirin and Vitamin D to Reduce COVID-19 Hospitalizations (LEAD COVID-19) (NCT04363840) is a randomized parallel assignment(Open Label) study with a start date of May 2020 and an end date of December 2020. The number of participants is 1080 and the study population are patients infected with COVID-19. The intervention group will receive either aspirin 81 mg once daily versus aspirin 81 mg once daily for 14 days plus a dietary supplement of 50,000 IU of vitamin D, to be taken orally once weekly for 2 weeks. The primary outcome is hospitalization for COVID-19 symptoms.
3) An Open Label Phase II Pilot Study of Hydroxychloroquine, Vitamin C, Vitamin D, and Zinc for the Prevention of COVID-19 Infection (HELPCOVID-19) (NCT04335084) is an open label trial, with assignment of participants to a single group, with start date April 2020 and end date July 2020. The number of participants is 600. The intervention is the use of hydroxychloroquine and a dietary supplement of vitamin C, vitamin D and zinc. The main outcome is the prevention of COVID-19 symptoms as recorded in a daily diary.
4) Impact of Zinc and Vitamin D3 Supplementation on the Survival of Aged Patients Infected with COVID-19 (ZnD3-CoVici) (NCT04351490) is a randomized open label parallel assignment trial with a start date April 2020 and end date July 2020. The number of participants is 3140 and the intervention is zinc gluconate capsule (15 mg x 2 per day) “25−OH-cholecalciferol drinkable solution 10 drops (2000 IU)” (precise dosing in microgram not mentioned) per day for 2 months. The main outcome is the survival rate in asymptomatic subjects at inclusion.
5) COVID-19 and Vitamin D Supplementation is a Multicenter Randomized Controlled Trial of High Dose Versus Standard Dose Vitamin D3 in High-risk COVID-19 Patients (CoVitTrial) (NCT04344041). It is an open label trial with random assignment and start date April 2020 and end date July 2020. The number of participants is 260 subjects with coronavirus infection. The intervention is either a single dose of cholecalciferol 400,000 IU compared to a single dose of 50,000 IU. The main outcome is number of deaths from any cause during the 14 days following inclusion and intervention.
6)WEST 2020D. Impact of vitamin D deficiency on prognosis of patients with novel coronavirus pneumonia (COVID-19). No additional information on this study is available but it looks to be an observational study.
7) Chinese Clinical Trial Registry. The relationship between Vitamin D and novel coronavirus pneumonia (COVID-19). More detailed information is missing but it seems to be an observational study.
8) In Spain, based on a pilot study carried out at the Reina Sofia University Hospital (Córdoba) in 76 hospitalized patients demonstrating a reduced ICU admission by more than 50 % among patients assigned to calcifediol treatment, a trial of calcifediol supplementation started on April 29th 2020. The trial is registered as “Prevention and treatment with Calcifediol of Coronavirus induced acute respiratory syndrome (SARS) COVID-19 (COVIDIOL)” (NCT04366908). It is a multi-center, randomized, open-label clinical trial to study the efficacy and safety of calcifediol in hospitalized patients with confirmed SARS-CoV-2 infection causing respiratory disease undergoing treatment with the best available therapy administering Calcifediol or not (calcifediol in soft capsules: 0.532 mg on the day of admission and 0.266 mg on day 3 and 7 and then weekly until discharge or ICU admission). Admission to Intensive Care Unit or death are the primary outcomes.
9. The Tehran University of Medical Sciences (Iran), in collaboration with the Boston University, is responsible for an Interventional Clinical Trial registered as “Preventive and Therapeutic Effects of Oral 25-hydroxyvitamin D3 on Coronavirus (COVID-19) in Adults (Oral 25-hydroxyvitamin D3 and COVID-19)”. It is a multicenter randomized double-blinded placebo-controlled clinical trial with parallel groups and allocation 1:1. (NCT04386850). All subjects in a stratified random sampling method based on age, sex, BMI and serum level of 25(OH)D (<10 ng/dL vs 10 to <20 ng/dL) with serum calcium < = 10.6 mg/dL will be recruited in the 25OHD3 or placebo group. Subjects in the case group will receive 25 mcg of 25OHD3 once daily at bedtime for 2 months and the control group will receive placebo daily for 2 months. Part of the study will deal with patients testing positive for COVID-19 and another arm of the study will evaluate the preventive potential of calcifediol by using the same treatment protocol for the health care providers and hospital workers with a negative test for COVID-19, or a close patient relative with a negative test for COVID-19 who lives with the infected patients. The Study Start Date was April 14th, 2020 with Estimated Primary Completion Date on November 15th, 2020, planning to enroll 1500 subjects.
In conclusion, coronavirus infection is a serious health problem awaiting an effective vaccine and/or antiviral treatment. The major complication of SARS-Covid-19 pneumonitis is ARDS mediated by a variety of mechanisms that may be aggravated by vitamin D deficiency and tapered down by activation of the vitamin D receptor. Several randomized clinical trials using either oral vitamin D or oral calcifediol (25OHD) are ongoing and should provide guidelines within a few months.
Author statement
All authors declare that they have not submitted the present MS elsewhere and declare no COI for this MS Roger Bouillon has received (small) lecture fees from Abiogen, Procter and Gamble, Fresenius, FAES farma and CERES.
Acknowledgements
This work was supported by grants COVID-011-2020 “Programa de Investigación clínica en COVID-19 de Andalucía”. Consejería de Salud y Familia. Fundación Progreso y Salud y Fundación para la Investigación Biomédica de Córdoba (FIBICO). Andalucía. Spain.
References
- 1.Bloch E.M., Shoham S., Casadevall A., Sachais B.S., Shaz B., Winters J.L., van Buskirk C., Grossman B.J., Joyner M., Henderson J.P., Pekosz A., Lau B., Wesolowski A., Katz L., Shan H., Auwaerter P.G., Thomas D., Sullivan D.J., Paneth N., Gehrie E., Spitalnik S., Hod E., Pollack L., Nicholson W.T., Pirofski L., Bailey J.A., Tobian A.A.R. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J. Clin. Invest. 2020 doi: 10.1172/jci138745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C., Hohmann E., Chu H.Y., Luetkemeyer A., Kline S., Lopez de Castilla D., Finberg R.W., Dierberg K., Tapson V., Hsieh L., Patterson T.F., Paredes R., Sweeney D.A., Short W.R., Touloumi G., Lye D.C., Ohmagari N., Oh M., Ruiz-Palacios G.M., Benfield T., Fätkenheuer G., Kortepeter M.G., Atmar R.L., Creech C.B., Lundgren J., Babiker A.G., Pett S., Neaton J.D., Burgess T.H., Bonnett T., Green M., Makowski M., Osinusi A., Nayak S., Lane H.C. Remdesivir for the treatment of Covid-19 — preliminary report. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2007764. NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bouillon R., Marcocci C., Carmeliet G., Bikle D., White J.H., Dawson-Hughes B., Lips P., Munns C.F., Lazaretti-Castro M., Giustina A., Bilezikian J. Skeletal and extraskeletal actions of vitamin d: current evidence and outstanding questions. Endocr. Rev. 2019;40:1109–1151. doi: 10.1210/er.2018-00126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martens P.-J., Gysemans C., Verstuyf A., Mathieu C. Vitamin d’s effect on immune function. Nutrients. 2020;12:1248. doi: 10.3390/nu12051248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bikle D.D., Patzek S., Wang Y. Physiologic and pathophysiologic roles of extra renal CYP27b1: case report and review. Bone Rep. 2018;8:255–267. doi: 10.1016/j.bonr.2018.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martineau A.R., Jolliffe D.A., Hooper R.L., Greenberg L., Aloia J.F., Bergman P., Dubnov-Raz G., Esposito S., Ganmaa D., Ginde A.A., Goodall E.C., Grant C.C., Griffiths C.J., Janssens W., Laaksi I., Manaseki-Holland S., Mauger D., Murdoch D.R., Neale R., Rees J.R., Simpson S., Stelmach I., Kumar G.T., Urashima M., Camargo C.A. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ. 2017;356 doi: 10.1136/bmj.i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hansdottir S., Monick M.M., Hinde S.L., Lovan N., Look D.C., Hunninghake G.W. Respiratory epithelial cells convert inactive vitamin d to its active form: potential effects on host defense. J. Immunol. 2008;181:7090–7099. doi: 10.4049/jimmunol.181.10.7090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., Tai Y., Bai C., Gao T., Song J., Xia P., Dong J., Zhao J., Wang F.S. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., Xia J., Yu T., Zhang X., Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bellani G., Laffey J.G., Pham T., Fan E., Brochard L., Esteban A., Gattinoni L., Van Haren F.M.P., Larsson A., McAuley D.F., Ranieri M., Rubenfeld G., Thompson B.T., Wrigge H., Slutsky A.S., Pesenti A. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA J. Am. Med. Assoc. 2016;315:788–800. doi: 10.1001/jama.2016.0291. [DOI] [PubMed] [Google Scholar]
- 12.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fan E., Brodie D., Slutsky A.S. Acute respiratory distress syndrome. JAMA. 2018;319:698. doi: 10.1001/jama.2017.21907. [DOI] [PubMed] [Google Scholar]
- 15.Standiford T.J., Ward P.A. Therapeutic targeting of acute lung injury and acute respiratory distress syndrome. Transl. Res. 2016;167:183–191. doi: 10.1016/j.trsl.2015.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Channappanavar R., Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cameron M.J., Bermejo-Martin J.F., Danesh A., Muller M.P., Kelvin D.J. Human immunopathogenesis of severe acute respiratory syndrome (SARS) Virus Res. 2008;133:13–19. doi: 10.1016/j.virusres.2007.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Min C.-K., Cheon S., Ha N.-Y., Sohn K.M., Kim Y., Aigerim A., Shin H.M., Choi J.-Y., Inn K.-S., Kim J.-H., Moon J.Y., Choi M.-S., Cho N.-H., Kim Y.-S. Comparative and kinetic analysis of viral shedding and immunological responses in MERS patients representing a broad spectrum of disease severity. Sci. Rep. 2016;6:25359. doi: 10.1038/srep25359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Williams A.E., Chambers R.C. The mercurial nature of neutrophils: still an enigma in ARDS? Am. J. Physiol. Lung Cell Mol. Physiol. 2014;306:L217–L230. doi: 10.1152/ajplung.00311.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Williams A.E., José R.J., Mercer P.F., Brealey D., Parekh D., Thickett D.R., O’Kane C., McAuley D.F., Chambers R.C. Evidence for chemokine synergy during neutrophil migration in ARDS. Thorax. 2017;72:66–73. doi: 10.1136/thoraxjnl-2016-208597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ichikawa A., Kuba K., Morita M., Chida S., Tezuka H., Hara H., Sasaki T., Ohteki T., Ranieri V.M., Dos Santos C.C., Kawaoka Y., Akira S., Luster A.D., Lu B., Penninger J.M., Uhlig S., Slutsky A.S., Imai Y. CXCL10-CXCR3 enhances the development of neutrophil-mediated fulminant lung injury of viral and nonviral origin. Am. J. Respir. Crit. Care Med. 2013;187:65–77. doi: 10.1164/rccm.201203-0508OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., Yang P., Sarao R., Wada T., Leong-Poi H., Crackower M.A., Fukamizu A., Hui C.C., Hein L., Uhlig S., Slutsky A.S., Jiang C., Penninger J.M. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Treml B., Neu N., Kleinsasser A., Gritsch C., Finsterwalder T., Geiger R., Schuster M., Janzek E., Loibner H., Penninger J., Loeckinger A. Recombinant angiotensin-converting enzyme 2 improves pulmonary blood flow and oxygenation in lipopolysaccharide-induced lung injury in piglets. Crit. Care Med. 2010;38:596–601. doi: 10.1097/CCM.0b013e3181c03009. [DOI] [PubMed] [Google Scholar]
- 24.Wenz M., Hoffmann B., Bohlender J., Kaczmarczyk G. Angiotensin II formation and endothelin clearance in ARDS patients in supine and prone positions. Intensive Care Med. 2000;26:292–298. doi: 10.1007/s001340051152. [DOI] [PubMed] [Google Scholar]
- 25.Kim J., Choi S.M., Lee J., Park Y.S., Lee C.H., Yim J.-J., Yoo C.-G., Kim Y.W., Han S.K., Lee S.-M. Effect of Renin-Angiotensin System Blockage in Patients with Acute Respiratory Distress Syndrome: A Retrospective Case Control Study. Korean J. Crit. Care Med. 2017;32:154–163. doi: 10.4266/kjccm.2016.00976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kuba K., Imai Y., Rao S., Jiang C., Penninger J.M. Lessons from SARS: control of acute lung failure by the SARS receptor ACE2. J. Mol. Med. 2006;84:814–820. doi: 10.1007/s00109-006-0094-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou P., Lou Yang X., Wang X.G., Hu B., Zhang L., Zhang W., Si H.R., Zhu Y., Li B., Huang C.L., Chen H.D., Chen J., Luo Y., Guo H., Di Jiang R., Liu M.Q., Chen Y., Shen X.R., Wang X., Zheng X.S., Zhao K., Chen Q.J., Deng F., Liu L.L., Yan B., Zhan F.X., Wang Y.Y., Xiao G.F., Shi Z.L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhao Y., Zhao Z., Wang Y., Zhou Y., Ma Y., Zuo W. Single-cell RNA expression profiling of ACE2, the putative receptor of Wuhan 2019-nCov. BioRxiv. 2020 doi: 10.1101/2020.01.26.919985. 2020.01.26.919985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Dagenais N.J., Jamali F. Protective effects of angiotensin II interruption: evidence for antiinflammatory actions. Pharmacotherapy. 2005;25:1213–1229. doi: 10.1592/phco.2005.25.9.1213. [DOI] [PubMed] [Google Scholar]
- 30.Patel V.B., Basu R., Oudit G.Y. ACE2/Ang 1-7 axis: a critical regulator of epicardial adipose tissue inflammation and cardiac dysfunction in obesity. Adipocyte. 2016;5:306–311. doi: 10.1080/21623945.2015.1131881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gouda M.M., Shaikh S.B., Bhandary Y.P. Inflammatory and fibrinolytic system in acute respiratory distress syndrome. Lung. 2018;196:609–616. doi: 10.1007/s00408-018-0150-6. [DOI] [PubMed] [Google Scholar]
- 32.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., Mehra M.R., Schuepbach R.A., Ruschitzka F., Moch H. Correspondence Endothelial cell infection and endotheliitis in. Lancet. 2020;6736:19–20. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Giannis D., Ziogas I.A., Gianni P. Coagulation disorders in coronavirus infected patients: COVID-19, SARS-CoV-1, MERS-CoV and lessons from the past. J. Clin. Virol. 2020;127 doi: 10.1016/j.jcv.2020.104362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bikdeli B., Madhavan M.V., Jimenez D., Chuich T., Dreyfus I., Driggin E., Der Nigoghossian C., Ageno W., Madjid M., Guo Y., Tang L.V., Hu Y., Giri J., Cushman M., Quéré I., Dimakakos E.P., Gibson C.M., Lippi G., Favaloro E.J., Fareed J., Caprini J.A., Tafur A.J., Burton J.R., Francese D.P., Wang E.Y., Falanga A., McLintock C., Hunt B.J., Spyropoulos A.C., Barnes G.D., Eikelboom J.W., Weinberg I., Schulman S., Carrier M., Piazza G., Beckman J.A., Steg P.G., Stone G.W., Rosenkranz S., Goldhaber S.Z., Parikh S.A., Monreal M., Krumholz H.M., Konstantinides S.V., Weitz J.I., Lip G.Y.H. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up. J. Am. Coll. Cardiol. 2020 doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang J., Chen L., Chen B., Meliton A., Liu S.Q., Shi Y., Liu T., Deb D.K., Solway J., Chun Li Y. Chronic Activation of the renin-angiotensin system induces lung fibrosis. Sci. Rep. 2015;5 doi: 10.1038/srep15561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ye Z., Zhang Y., Wang Y., Huang Z., Song B. Chest CT manifestations of new coronavirus disease 2019 (COVID-19): a pictorial review. Eur. Radiol. 2020:1–9. doi: 10.1007/s00330-020-06801-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xu J., Yang J., Chen J., Luo Q., Zhang Q., Zhang H. Vitamin D alleviates lipopolysaccharide-induced acute lung injury via regulation of the renin-angiotensin system. Mol. Med. Rep. 2017;16:7432–7438. doi: 10.3892/mmr.2017.7546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shi Y.Y., Liu T.J., Fu J.H., Xu W., Wu L.L., Hou A.N., Xue X.D. Vitamin D/VDR signaling attenuates lipopolysaccharide-induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier. Mol. Med. Rep. 2016;13:1186–1194. doi: 10.3892/mmr.2015.4685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kong J., Zhu X., Shi Y., Liu T., Chen Y., Bhan I., Zhao Q., Thadhani R., Chun Li Y. VDR attenuates acute lung injury by blocking Ang-2-Tie-2 pathway and renin-angiotensin system. Mol. Endocrinol. 2013;27:2116–2125. doi: 10.1210/me.2013-1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zheng S., Yang J., Hu X., Li M., Wang Q., Dancer R.C.A., Parekh D., Gao-Smith F., Thickett D.R., Jin S. Vitamin D attenuates lung injury via stimulating epithelial repair, reducing epithelial cell apoptosis and inhibits TGF-β induced epithelial to mesenchymal transition. Biochem. Pharmacol. 2020:113955. doi: 10.1016/j.bcp.2020.113955. [DOI] [PubMed] [Google Scholar]
- 41.Ishii M., Yamaguchi Y., Isumi K., Ogawa S., Akishita M. Transgenic Mice Overexpressing Vitamin D Receptor (VDR) Show Anti-Inflammatory Effects in Lung Tissues. Inflammation. 2017;40:2012–2019. doi: 10.1007/s10753-017-0641-2. [DOI] [PubMed] [Google Scholar]
- 42.Rafique A., Rejnmark L., Heickendorff L., Møller H.J. 25(OH)D 3 and 1.25(OH) 2 D 3 inhibits TNF-α expression in human monocyte derived macrophages. PLoS One. 2019;14 doi: 10.1371/journal.pone.0215383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Andrukhov O., Andrukhova O., Hulan U., Tang Y., Bantleon H.P., Rausch-Fan X. Both 25-hydroxyvitamin-D3 and 1,25-dihydroxyvitamin- D3 reduces inflammatory response in human periodontal ligament cells. PLoS One. 2014;9:e90301. doi: 10.1371/journal.pone.0090301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bischoff-Ferrari H.A. Optimal serum 25-hydroxyvitamin D levels for multiple health outcomes. Adv. Exp. Med. Biol. 2014;810:500–525. doi: 10.1007/978-1-4939-0437-2_28. [DOI] [PubMed] [Google Scholar]
- 45.Selvaraj P., Harishankar M., Singh B., Banurekha V.V., Jawahar M.S. Effect of vitamin D 3 on chemokine expression in pulmonary tuberculosis. Cytokine. 2012;60:212–219. doi: 10.1016/j.cyto.2012.06.238. [DOI] [PubMed] [Google Scholar]
- 46.Scolletta S., Colletti M., Di Luigi L., Crescioli C. Vitamin D receptor agonists target CXCL10: new therapeutic tools for resolution of inflammation. Mediators Inflamm. 2013;2013 doi: 10.1155/2013/876319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kong J., Zhang Z., Musch M.W., Ning G., Sun J., Hart J., Bissonnette M., Yan C.L. Novel role of the vitamin D receptor in maintaining the integrity of the intestinal mucosal barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2007;294 doi: 10.1152/ajpgi.00398.2007. [DOI] [PubMed] [Google Scholar]
- 48.Takano Y., Mitsuhashi H., Ueno K. 1α,25-Dihydroxyvitamin D 3 inhibits neutrophil recruitment in hamster model of acute lung injury. Steroids. 2011;76:1305–1309. doi: 10.1016/j.steroids.2011.06.009. [DOI] [PubMed] [Google Scholar]
- 49.Li Y.C., Qiao G., Uskokovic M., Xiang W., Zheng W., Kong J. Vitamin D: a negative endocrine regulator of the renin-angiotensin system and blood pressure. J. Steroid Biochem. Mol. Biol. 2004:387–392. doi: 10.1016/j.jsbmb.2004.03.004. [DOI] [PubMed] [Google Scholar]
- 50.Zhou C., Lu F., Cao K., Xu D., Goltzman D., Miao D. Calcium-independent and 1,25(OH)2D3-dependent regulation of the renin-angiotensin system in 1α-hydroxylase knockout mice. Kidney Int. 2008;74:170–179. doi: 10.1038/ki.2008.101. [DOI] [PubMed] [Google Scholar]
- 51.Chen L.N., Yang X.H., Nissen D.H., Chen Y.Y., Wang L.J., Wang J.H., Gao J.L., Zhang L.Y. Dysregulated renin-Angiotensin system contributes to acute lung injury caused by hind-limb ischemia-reperfusion in mice. Shock. 2013;40:420–429. doi: 10.1097/SHK.0b013e3182a6953e. [DOI] [PubMed] [Google Scholar]
- 52.Liu D., Fang Y.X., Wu X., Tan W., Zhou W., Zhang Y., Liu Y.Q., Li G.Q. 1,25-(OH)2D3/Vitamin D receptor alleviates systemic lupus erythematosus by downregulating Skp2 and upregulating p27, Cell Commun. Signal. 2019;17 doi: 10.1186/s12964-019-0488-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yang E.S., Burnstein K.L. Vitamin d inhibits G1 to S progression in LNCaP prostate Cancer cells through p27Kip1 stabilization and Cdk2 mislocalization to the cytoplasm. J. Biol. Chem. 2003;278:46862–46868. doi: 10.1074/jbc.M306340200. [DOI] [PubMed] [Google Scholar]
- 54.Gassen N.C., Niemeyer D., Muth D., Corman V.M., Martinelli S., Gassen A., Hafner K., Papies J., Mösbauer K., Zellner A., Zannas A.S., Herrmann A., Holsboer F., Brack-Werner R., Boshart M., Müller-Myhsok B., Drosten C., Müller M.A., Rein T. SKP2 attenuates autophagy through Beclin1-ubiquitination and its inhibition reduces MERS-Coronavirus infection. Nat. Commun. 2019;10 doi: 10.1038/s41467-019-13659-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fernández Á.F., Sebti S., Wei Y., Zou Z., Shi M., McMillan K.L., He C., Ting T., Liu Y., Chiang W.C., Marciano D.K., Schiattarella G.G., Bhagat G., Moe O.W., Hu M.C., Levine B. Disruption of the beclin 1-BCL2 autophagy regulatory complex promotes longevity in mice. Nature. 2018;558:136–140. doi: 10.1038/s41586-018-0162-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Andersen K.G., Rambaut A., Lipkin W.I., Holmes E.C., Garry R.F. The proximal origin of SARS-CoV-2. Nat. Med. 2020;26:450–452. doi: 10.1038/s41591-020-0820-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Talreja H., Tan J., Dawes M., Supershad S., Rabindranath K., Fisher J., Valappil S., van der Merwe V., Wong L., van der Merwe W., Paton J. A consensus statement on the use of angiotensin receptor blockers and angiotensin converting enzyme inhibitors in relation to COVID-19 (corona virus disease 2019) N. Z. Med. J. 2020;133:85–87. http://www.ncbi.nlm.nih.gov/pubmed/32242182 (accessed May 2, 2020) [PubMed] [Google Scholar]
- 58.Guo J., Huang Z., Lin L., Lv J. Coronavirus disease 2019 (COVID-19) and cardiovascular disease: a viewpoint on the potential influence of angiotensin-converting enzyme Inhibitors/Angiotensin receptor blockers on onset and severity of severe acute respiratory syndrome coronavirus 2 infection. J. Am. Heart Assoc. 2020;9 doi: 10.1161/JAHA.120.016219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vaduganathan M., Vardeny O., Michel T., McMurray J.J.V., Pfeffer M.A., Solomon S.D. Renin–Angiotensin–Aldosterone system inhibitors in patients with Covid-19. N. Engl. J. Med. 2020 doi: 10.1056/nejmsr2005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lubel J., Garg M. Renin–Angiotensin–Aldosterone system inhibitors in Covid-19. N. Engl. J. Med. 2020;382 doi: 10.1056/NEJMc2013707. NEJMc2013707. [DOI] [PubMed] [Google Scholar]
- 61.Brinkmann V., Reichard U., Goosmann C., Fauler B., Uhlemann Y., Weiss D.S., Weinrauch Y., Zychlinsky A. Neutrophil extracellular traps kill Bacteria. Science (80-.) 2004;303:1532–1535. doi: 10.1126/science.1092385. [DOI] [PubMed] [Google Scholar]
- 62.Barnes B.J., Adrover J.M., Baxter-Stoltzfus A., Borczuk A., Cools-Lartigue J., Crawford J.M., Daßler-Plenker J., Guerci P., Huynh C., Knight J.S., Loda M., Looney M.R., McAllister F., Rayes R., Renaud S., Rousseau S., Salvatore S., Schwartz R.E., Spicer J.D., Yost C.C., Weber A., Zuo Y., Egeblad M. Targeting potential drivers of COVID-19: neutrophil extracellular traps. J. Exp. Med. 2020;217 doi: 10.1084/jem.20200652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Agraz-Cibrian J.M., Giraldo D.M., Urcuqui-Inchima S. 1,25-Dihydroxyvitamin D3 induces formation of neutrophil extracellular trap-like structures and modulates the transcription of genes whose products are neutrophil extracellular trap-associated proteins: a pilot study. Steroids. 2019;141:14–22. doi: 10.1016/j.steroids.2018.11.001. [DOI] [PubMed] [Google Scholar]
- 64.Hansdottir S., Monick M.M., Lovan N., Powers L., Gerke A., Hunninghake G.W. Vitamin d decreases respiratory syncytial virus induction of NF-κB–Linked chemokines and cytokines in airway epithelium while maintaining the antiviral state. J. Immunol. 2010;184:965–974. doi: 10.4049/jimmunol.0902840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen H., Lu R., guo Zhang Y., Sun J. Vitamin d receptor deletion leads to the destruction of tight and adherens junctions in lungs. Tissue Barriers. 2018;6:1–13. doi: 10.1080/21688370.2018.1540904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kose M., Bastug O., Sonmez M.F., Per S., Ozdemir A., Kaymak E., Yahşi H., Ozturk M.A. Protective effect of vitamin D against hyperoxia-induced lung injury in newborn rats. Pediatr. Pulmonol. 2017;52:69–76. doi: 10.1002/ppul.23500. [DOI] [PubMed] [Google Scholar]
- 67.Margetic S. Inflammation and hemostasis. Biochem. Medica. 2012:49–62. doi: 10.11613/bm.2012.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ohsawa M., Koyama T., Yamamoto K., Hirosawa S., Kamei S., Kamiyama R. 1α,25-Dihydroxyvitamin D3 and its potent synthetic analogs downregulate tissue factor and upregulate thrombomodulin expression in monocytic cells, counteracting the effects of tumor necrosis factor and oxidized LDL. Circulation. 2000;102:2867–2872. doi: 10.1161/01.CIR.102.23.2867. [DOI] [PubMed] [Google Scholar]
- 69.Aihara K.I., Azuma H., Akaike M., Ikeda Y., Yamashita M., Sudo T., Hayashi H., Yamada Y., Endoh F., Fujimura M., Yoshida T., Yamaguchi H., Hashizume S., Kato M., Yoshimura K., Yamamoto Y., Kato S., Matsumoto T. Disruption of nuclear vitamin D receptor gene causes enhanced thrombogenicity in mice. J. Biol. Chem. 2004;279:35798–35802. doi: 10.1074/jbc.M404865200. [DOI] [PubMed] [Google Scholar]
- 70.Martinez-Moreno J.M., Herencia C., De Oca A.M., Muñoz-Castañeda J.R., Rodríguez-Ortiz M.E., Diáz-Tocados J.M., Peralbo-Santaella E., Camargo A., Canalejo A., Rodriguez M., Velasco-Gimena F., Almaden Y. Vitamin D modulates tissue factor and protease-activated receptor 2 expression in vascular smooth muscle cells. FASEB J. 2016;30:1367–1376. doi: 10.1096/fj.15-272872. [DOI] [PubMed] [Google Scholar]
- 71.Wu W.X., He D.R. Low vitamin d levels are associated with the development of deep venous thromboembolic events in patients with ischemic stroke. Clin. Appl. Thromb. Hemost. 2018;24:69S–75S. doi: 10.1177/1076029618786574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Topaloglu O., Arslan M.S., Karakose M., Ucan B., Ginis Z., Cakir E., Akkaymak E.T., Sahin M., Ozbek M., Cakal E., Delibasi T. Is There Any Association Between Thrombosis and Tissue Factor Pathway Inhibitor Levels in Patients With Vitamin D Deficiency? Clin. Appl. Thromb. Hemost. 2015;21:428–433. doi: 10.1177/1076029613509477. [DOI] [PubMed] [Google Scholar]
- 73.Holick M.F. Medical progress: vitamin d deficiency. N. Engl. J. Med. 2007;357:266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 74.Sluyter J.D., Camargo C.A., Waayer D., Lawes C.M.M., Toop L., Khaw K.T., Scragg R. Effect of monthly, high-dose, long-term vitamin D on lung function: a randomized controlled trial. Nutrients. 2017;9 doi: 10.3390/nu9121353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bouillon R. Vitamin D status in Africa is worse than in other continents. Lancet Glob. Heal. 2020;8:e20–e21. doi: 10.1016/S2214-109X(19)30492-9. [DOI] [PubMed] [Google Scholar]
- 76.Dancer R.C.A., Parekh D., Lax S., D’Souza V., Zheng S., Bassford C.R., Park D., Bartis D.G., Mahida R., Turner A.M., Sapey E., Wei W., Naidu B., Stewart P.M., Fraser W.D., Christopher K.B., Cooper M.S., Gao F., Sansom D.M., Martineau A.R., Perkins G.D., Thickett D.R. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS) Thorax. 2015;70:617–624. doi: 10.1136/thoraxjnl-2014-206680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Thickett D.R., Moromizato T., Litonjua A.A., Amrein K., Quraishi S.A., Lee-Sarwar K.A., Mogensen K.M., Purtle S.W., Gibbons F.K., Camargo C.A., Giovannucci E., Christopher K.B. Association between prehospital vitamin D status and incident acute respiratory failure in critically ill patients: a retrospective cohort study. BMJ Open Respir. Res. 2015;2:1–8. doi: 10.1136/bmjresp-2014-000074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Park S., Lee M.G., Hong S.B., Lim C.M., Koh Y., Huh J.W. Effect of vitamin D deficiency in korean patients with acute respiratory distress syndrome. Korean J. Intern. Med. 2018;33:1129–1136. doi: 10.3904/kjim.2017.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.http://www.chictr.org.cn/showprojen.aspx?proj=52006, Impact of vitamin D deficiency on prognosis of patients with novel coronavirus pneumonia (COVID-19), (2018) 3–4. http://www.chictr.org.cn/showprojen.aspx?proj=49302 (accessed May 2, 2020).
- 80.http://www.chictr.org.cn/showprojen.aspx?proj=52006, The relationship between Vitamin D andnovel coronavirus pneumonia (COVID-19), (2018) 3–4. http://www.chictr.org.cn/showprojen.aspx?proj=51390 (accessed May 2, 2020).
- 81.Martucci G., McNally D., Parekh D., Zajic P., Tuzzolino F., Arcadipane A., Christopher K.B., Dobnig H., Amrein K. Trying to identify who may benefit most from future vitamin D intervention trials: a post hoc analysis from the VITDAL-ICU study excluding the early deaths. Crit. Care. 2019;23:200. doi: 10.1186/s13054-019-2472-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Bouillon R., Bikle D. Vitamin d metabolism revised: fall of dogmas. J. Bone Miner. Res. 2019;34:1985–1992. doi: 10.1002/jbmr.3884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jolliffe D.A., Stefanidis C., Wang Z., Kermani N.Z., Dimitrov V., White J.H., McDonough J.E., Janssens W., Pfeffer P., Griffiths C.J., Bush A., Guo Y., Christenson S., Adcock I.M., Chung K.F., Thummel K.E., Martineau A.R. Vitamin d metabolism is dysregulated in asthma and chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2020 doi: 10.1164/rccm.201909-1867oc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Quesada-Gomez J.M., Bouillon R. Is calcifediol better than cholecalciferol for vitamin D supplementation? Osteoporos. Int. 2018;29:1697–1711. doi: 10.1007/s00198-018-4520-y. [DOI] [PubMed] [Google Scholar]
- 85.Grein J., Ohmagari N., Shin D., Diaz G., Asperges E., Castagna A., Feldt T., Green G., Green M.L., Lescure F.-X., Nicastri E., Oda R., Yo K., Quiros-Roldan E., Studemeister A., Redinski J., Ahmed S., Bernett J., Chelliah D., Chen D., Chihara S., Cohen S.H., Cunningham J., D’Arminio Monforte A., Ismail S., Kato H., Lapadula G., L’Her E., Maeno T., Majumder S., Massari M., Mora-Rillo M., Mutoh Y., Nguyen D., Verweij E., Zoufaly A., Osinusi A.O., DeZure A., Zhao Y., Zhong L., Chokkalingam A., Elboudwarej E., Telep L., Timbs L., Henne I., Sellers S., Cao H., Tan S.K., Winterbourne L., Desai P., Mera R., Gaggar A., Myers R.P., Brainard D.M., Childs R., Flanigan T. Compassionate use of remdesivir for patients with severe Covid-19. N. Engl. J. Med. 2020 doi: 10.1056/nejmoa2007016. [DOI] [PMC free article] [PubMed] [Google Scholar]