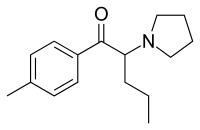
Pyrovalerone
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.230.426 |
| Chemical and physical data | |
| Formula | C16H23NO |
| Molar mass | 245.366 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Pyrovalerone (Centroton, 4-Methyl-β-keto-prolintane, Thymergix, O-2371)[2] is a central nervous system (CNS) stimulant that acts as a norepinephrine–dopamine reuptake inhibitor (NDRI). It was developed in the 1980s and had briefly been approved in Spain and France for chronic fatigue or lethargy[3] and as an appetite suppressant, but was withdrawn from both markets around 2001 due to safety concerns including problems with abuse and dependence.[4] It is closely related on a structural level to a number of other cathinone stimulants, such as α-PVP, MDPV and prolintane.
Side effects of pyrovalerone include decreased appetite, anxiety, fragmented sleep or insomnia, and trembling, shaking, or muscle tremors. Withdrawal symptoms following abuse upon discontinuation often results in depression.
The R-enantiomer of pyrovalerone is devoid of pharmacologic activity.[5]
See also
[edit]- 4-Et-PVP
- α-Pyrrolidinohexiophenone (α-PHP)
- α-Pyrrolidinopentiothiophenone (α-PVT)
- Methylenedioxypyrovalerone (MDPV)
- Naphyrone (O-2482)
- Prolintane (Promotil, Katovit)
- 4'-Methyl-α-pyrrolidinohexiophenone (MPHP, 4-MPHP)
References
[edit]- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ US Patent 3314970
- ^ Gardos G, Cole JO (October 1971). "Evaluation of pyrovalerone in chronically fatigued volunteers". Current Therapeutic Research, Clinical and Experimental. 13 (10): 631–5. PMID 4402508.
- ^ Deniker P, Lôo H, Cuche H, Roux JM (November 1975). "[Abuse of pyrovalerone by drug addicts]". Annales médico-psychologiques. 2 (4): 745–8. PMID 9895.
- ^ Meltzer PC, Butler D, Deschamps JR, Madras BK (February 2006). "1-(4-Methylphenyl)-2-pyrrolidin-1-yl-pentan-1-one (Pyrovalerone) analogues: a promising class of monoamine uptake inhibitors". Journal of Medicinal Chemistry. 49 (4): 1420–32. doi:10.1021/jm050797a. PMC 2602954. PMID 16480278.

