From Wikipedia, the free encyclopedia
Chemical compound
Pharmaceutical compound
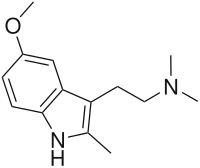
5-Methoxy-2,N,N-trimethyltryptamine (5-MeO-2,N,N-TMT, 5-MeO-TMT) is a psychoactive drug of the tryptamine chemical class which acts as a psychedelic. It was first synthesized by Alexander Shulgin and reported in his book TiHKAL ("Tryptamines i Have Known And Loved").[1] 5-MeO-TMT is claimed to show psychoactive effects at a dosage of 75–150 mg orally, but these are relatively mild compared to those of other similar compounds. This suggests that while the methyl group on the 2-position of the molecule has impaired the binding of metabolic enzymes like monoamine oxidase (MAO), it is also interfering with binding to and/or activation of the serotonin 5-HT2A receptor, the target responsible for mediating the hallucinogenic effects of such compounds.
|
|---|
| Tryptamines |
- 1-Methyl-T
- 1-Methylpsilocin
- 2-HO-NMT
- 2-Me-DET
- 2-Methyl-5-HT
- 2,N,N-TMT
- 4,5-DHP-DMT
- 4-AcO-DALT
- 4-AcO-DET
- 4-AcO-DiPT
- 4-AcO-DPT
- 4-AcO-EPT
- 4-AcO-MALT
- 4-AcO-MET
- 4-AcO-MiPT
- 4-AcO-NMT
- 4-AcO-TMT
- 4-F-5-MeO-DMT
- 4-Fluoro-T
- 4-HO-5-MeO-DMT
- 4-HO-DALT
- 4-HO-DBT
- 4-HO-DET
- 4-HO-DiPT
- 4-HO-DPT
- 4-HO-DSBT
- 4-HO-EPT
- 4-HO-MALT
- 4-HO-MET
- 4-HO-McPT
- 4-HO-McPeT
- 4-HO-MiPT
- 4-HO-MPT
- 4-HO-MsBT
- 4-HO-NALT
- 4-HO-NMT
- 4-HO-PiPT
- 4-HO-pyr-T
- 4-HO-TMT
- 4-HT
- 4-MeO-DiPT
- 4-MeO-DMT
- 4-MeO-MiPT
- 4-MeO-T
- 4-PrO-DMT
- 4,5-MDO-DMT
- 4,5-MDO-DiPT
- 5-BT
- 5-Bromo-DMT
- 5-Bromo-T
- 5-Chloro-DMT
- 5-Chloro-T
- 5-CT
- 5-Ethoxy-DMT
- 5-Ethyl-DMT
- 5-Fluoro-DET
- 5-Fluoro-DMT
- 5-Fluoro-EPT
- 5-Fluoro-MET
- 5-Fluoro-T
- 5-HO-DiPT
- 5-HTP (oxitriptan)
- 5-MeO-2-TMT
- 5-MeO-34MPEMT
- 5-MeO-7,N,N-TMT
- 5-MeO-DALT
- 5-MeO-DBT
- 5-MeO-DET
- 5-MeO-DiPT
- 5-MeO-DMT
- 5-MeO-DPT
- 5-MeO-EiPT
- 5-MeO-EPT
- 5-MeO-MALT
- 5-MeO-MET
- 5-MeO-MiPT
- 5-MeO-NET
- 5-MeO-NiPT
- 5-MeO-NMT
- 5-MeO-pyr-T
- 5-MeO-NBpBrT
- 5-MeO-T (5-MT; mexamine)
- 5-MeO-T-NBOMe
- 5-MeS-DMT
- 5-Methyl-DMT
- 5-Methyl-T
- 5-MT-NB3OMe
- 5-NOT
- 5,6-MeO-MiPT
- 5,6-MDO-DiPT
- 5,6-MDO-DMT
- 5,6-MDO-MiPT
- 5,6-DHT
- 5,7-DHT
- 6-Fluoro-DMT
- 6-Fluoro-T
- 6-MeO-DMT
- 6-MeO-T
- 6-Methyl-T
- 7-Chloro-T
- 7-MeO-T
- 7-Methyl-DMT
- 7-Methyl-T
- Acetryptine (5-AT)
- Aeruginascin (4-PO-TMT)
- AGH-107
- AGH-192
- AH-494
- ALiPT
- Alpertine
- Baeocystin (4-PO-NMT)
- Benzotript (4-chlorobenzoyl-L-tryptophan)
- Bufotenidine (5-HTQ)
- Bufotenin (5-HO-DMT)
- Convolutindole A
- CP-132,484
- DALT
- DBT
- Desformylflustrabromine
- DET
- DiPT
- DMT
- DPT
- E-6801
- E-6837
- EiPT
- EMDT
- EPT
- Ethocybin (4-PO-DET)
- FGIN-127
- FGIN-143
- FT-104
- HIOC
- Idalopirdine
- Indolylethylfentanyl
- Indorenate
- Iprocin (4-HO-DiPT)
- Isamide
- Lespedamine
- MBT
- MET
- Milipertine
- Miprocin (4-HO-MiPT)
- MiPT
- MPT
- MS-245
- MSBT
- N-Feruloylserotonin (moschamine)
- NET/NETP
- NiPT
- NMT
- Norbaeocystin (4-PO-T)
- NTBT
- O-4310
- O-Pivalylbufotenine
- Oxypertine
- PiPT
- Psilacetin (O-acetylpsilocin; 4-AcO-DMT)
- Psilocin (4-HO-DMT)
- Psilocybin (4-PO-DMT)
- Pyr-T
- RS134-49
- Serotonin (5-HT)
- Solypertine
- ST-1936
- Tryptamine (T)
- Tryptophan
- Yuremamine
- Z2876442907
|
|---|
| N-Acetyltryptamines | |
|---|
| α-Alkyltryptamines |
- 2,α-DMT
- 4-HO-αMT
- 4-HO-MPMI (lucigenol)
- 4-Me-αET
- 4-Me-αMT
- 5-Chloro-αET
- 5-Chloro-αMT
- 5-Ethoxy-αMT
- 5-Fluoro-αET
- 5-Fluoro-αMT
- 5-iPrO-αMT
- 5-MeO-α,N,N-TMT
- 5-MeO-αET
- 5-MeO-αMT
- 5-MeO-MPMI
- 5-Methyl-αET
- 6-Fluoro-αMT
- 7-Chloro-αMT
- 7-Methyl-αET
- α-Methyl-5-HTP
- α-Methylmelatonin
- α-Methylserotonin (5-HO-αMT)
- α-Methyltryptophan (αMTP)
- α,N-DMT (N-methyl-αMT)
- α,N,N-TMT
- α,N,O-TMS
- αET (etryptamine)
- αMT
- AL-37350A (4,5-DHP-αMT)
- BK-5Br-NM-AMT
- BK-5Cl-NM-AMT
- BK-5F-NM-AMT
- BK-NM-AMT
- BNC-210
- BW-723C86
- CP-135807
- IPAP (α,N-DPT)
- MPMI
|
|---|
| Triptans | |
|---|
| Cyclized tryptamines |
- Bay R 1531
- Ciclindole
- Cyclic 3-OHM
- Ergolines and lysergamides (e.g., LSD)
- Flucindole
- Harmala alkaloids and β-carbolines (e.g., 6-MeO-THH, 9-Me-BC, β-carboline (norharman), harmaline, harmalol, harmane, harmine, pinoline, tetrahydroharmine, tryptoline)
- Iboga alkaloids and related (e.g., DM-506 (ibogaminalog), ibogaine, ibogamine, noribogaine, tabernanthalog, tabernanthine)
- LY-266,097
- Metralindole
- NDTDI
- PHA-57378
- PNU-22394
- PNU-181731
- RU-28306
- Yohimbans (e.g., yohimbine, rauwolscine, spegatrine, corynanthine, ajmalicine, reserpine, deserpidine, rescinnamine)
|
|---|
| Isotryptamines | |
|---|
| Related compounds | |
|---|
|
|---|
Psychedelics
(5-HT2A
agonists) | | Benzofurans | |
|---|
Lyserg‐
amides | |
|---|
Phenethyl‐
amines | | 2C-x |
|
|---|
| 3C-x | |
|---|
| 4C-x | |
|---|
| DOx | |
|---|
| HOT-x | |
|---|
| MDxx | |
|---|
| Mescaline (subst.) | |
|---|
| TMAs |
- TMA
- TMA-2
- TMA-3
- TMA-4
- TMA-5
- TMA-6
|
|---|
| Others | |
|---|
|
|---|
| Piperazines | |
|---|
| Tryptamines | | alpha-alkyltryptamines | |
|---|
| x-DALT | |
|---|
| x-DET | |
|---|
| x-DiPT | |
|---|
| x-DMT |
- 4,5-DHP-DMT
- 2,N,N-TMT
- 4-AcO-DMT
- 4-HO-5-MeO-DMT
- 4,N,N-TMT
- 4-Propionyloxy-DMT
- 5,6-diBr-DMT
- 5-AcO-DMT
- 5-Bromo-DMT
- 5-MeO-2,N,N-TMT
- 5-MeO-4,N,N-TMT
- 5-MeO-α,N,N-TMT
- 5-MeO-DMT
- 5-N,N-TMT
- 7,N,N-TMT
- α,N,N-TMT
- (Bufotenin) 5-HO-DMT
- DMT
- Norbaeocystin
- (Psilocin) 4-HO-DMT
- (Psilocybin) 4-PO-DMT
|
|---|
| x-DPT | |
|---|
| Ibogaine-related | |
|---|
| x-MET | |
|---|
| x-MiPT | |
|---|
| Others | |
|---|
|
|---|
| Others | |
|---|
|
|---|
Dissociatives
(NMDAR
antagonists) | |
|---|
Deliriants
(mAChR
antagonists) | |
|---|
| Others | |
|---|