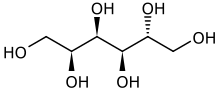
Sorbitol (/ˈsɔː(r)bɪtɒl/), less commonly known as glucitol (/ˈɡluːsɪtɒl/), is a sugar alcohol with a sweet taste which the human body metabolizes slowly. It can be obtained by reduction of glucose, which changes the converted aldehyde group (−CHO) to a primary alcohol group (−CH2OH). Most sorbitol is made from potato starch, but it is also found in nature, for example in apples, pears, peaches, and prunes.[4] It is converted to fructose by sorbitol-6-phosphate 2-dehydrogenase. Sorbitol is an isomer of mannitol, another sugar alcohol; the two differ only in the orientation of the hydroxyl group on carbon 2.[5] While similar, the two sugar alcohols have very different sources in nature, melting points, and uses.
| |

| |
| Names | |
|---|---|
| IUPAC name
D-Glucitol[1]
| |
| Systematic IUPAC name
(2S,3R,4R,5R)-Hexane-1,2,3,4,5,6-hexol | |
| Other names
D-Sorbitol; Sorbogem; Sorbo
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.056 |
| E number | E420 (thickeners, ...) |
| KEGG | |
| MeSH | Sorbitol |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C6H14O6 | |
| Molar mass | 182.17 g/mol |
| Appearance | White crystalline powder |
| Density | 1.49 g/cm3[2] |
| Melting point | 94–96 °C (201–205 °F; 367–369 K)[2] |
| 2350 g/L[2] | |
| log P | -4.67[3] |
| -107.80·10−6 cm3/mol | |
| Pharmacology | |
| A06AD18 (WHO) A06AG07 (WHO) B05CX02 (WHO) V04CC01 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | > 100 °C (212 °F; 373 K)[2] |
| 420 °C (788 °F; 693 K)[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
As an over-the-counter drug, sorbitol is used as a laxative to treat constipation.[6]
Synthesis
editSorbitol may be synthesised via a glucose reduction reaction[7] in which the converted aldehyde group is converted into a hydroxyl group. The reaction requires NADH and is catalyzed by aldose reductase. Glucose reduction is the first step of the polyol pathway of glucose metabolism, and is implicated in multiple diabetic complications.
The mechanism involves a tyrosine residue in the active site of aldehyde reductase. The hydrogen atom on NADH is transferred to the electrophilic aldehyde carbon atom; electrons on the aldehyde carbon-oxygen double bond are transferred to the oxygen that abstracts the proton on tyrosine side chain to form the hydroxyl group. The role of aldehyde reductase tyrosine phenol group is to serve as a general acid to provide proton to the reduced aldehyde oxygen on glucose.
Glucose reduction is not the major glucose metabolism pathway in a normal human body, where the glucose level is in the normal range. However, in diabetic patients whose blood glucose level is high, up to 1/3 of their glucose could go through the glucose reduction pathway. This will consume NADH and eventually leads to cell damage.
Uses
editSweetener
editSorbitol is a sugar substitute, and when used in food it has the INS number and E number 420. Sorbitol is about 60% as sweet as sucrose (table sugar).[8]
Sorbitol is referred to as a nutritive sweetener because it provides some dietary energy. It is partly absorbed from the small intestine and metabolized in the body, and partly fermented in the large intestine. The fermentation produces short-chain fatty acids, acetic acid, propionic acid, and butyric acid, which are mostly absorbed and provide energy, but also carbon dioxide, methane, and hydrogen which do not provide energy. Even though the heat of combustion of sorbitol is higher than that of glucose (having two extra hydrogen atoms), the net energy contribution is between 2.5 and 3.4 kilocalories per gram, versus the approximately 4 kilocalories (17 kilojoules) for carbohydrates.[9] It is often used in diet foods (including diet drinks and ice cream), mints, cough syrups, and sugar-free chewing gum.[10] Most bacteria cannot use sorbitol for energy, but it can be slowly fermented in the mouth by Streptococcus mutans, a bacterium that causes tooth decay. In contrast, many other sugar alcohols such as isomalt and xylitol are considered non-acidogenic.[11][12]
It also occurs naturally in many stone fruits and berries from trees of the genus Sorbus.[4][13]
Medical applications
editLaxative
editAs is the case with other sugar alcohols, foods containing sorbitol can cause gastrointestinal distress. Sorbitol can be used as a laxative when taken orally or as an enema.[6] Sorbitol works as a laxative by drawing water into the large intestine, stimulating bowel movements.[6][14] Sorbitol has been determined safe for use by the elderly, although it is not recommended without the advice of a physician.[6][15]
Sorbitol is commonly used orally as a one-time dose of 30–150 millilitres (1.1–5.3 imp fl oz; 1.0–5.1 US fl oz) 70% solution.[6] It may also be used as a one-time rectal enema.[6]
Other medical applications
editSorbitol is used in bacterial culture media to distinguish the pathogenic Escherichia coli O157:H7 from most other strains of E. coli, because it is usually unable to ferment sorbitol, unlike 93% of known E. coli strains.[16]
A treatment for hyperkalaemia (elevated blood potassium) uses sorbitol and the ion-exchange resin sodium polystyrene sulfonate (tradename Kayexalate).[17] The resin exchanges sodium ions for potassium ions in the bowel, while sorbitol helps to eliminate it. In 2010, the U.S. FDA issued a warning of increased risk for gastrointestinal necrosis with this combination.[18]
Sorbitol is also used in the manufacture of softgel capsules to store single doses of liquid medicines.[19]
Health care, food, and cosmetic uses
editSorbitol often is used in modern cosmetics as a humectant and thickener.[20] It is also used in mouthwash and toothpaste. Some transparent gels can be made only with sorbitol, because of its high refractive index.
Sorbitol is used as a cryoprotectant additive (mixed with sucrose and sodium polyphosphates) in the manufacture of surimi, a processed fish paste.[21] It is also used as a humectant in some cigarettes.[22]
Beyond its use as a sugar substitute in reduced-sugar foods, sorbitol is also used as a humectant in cookies and low-moisture foods like peanut butter and fruit preserves.[23] In baking, it is also valuable because it acts as a plasticizer, and slows down the staling process.[23]
Miscellaneous uses
editA mixture of sorbitol and potassium nitrate has found some success as an amateur solid rocket fuel. It has similar performance to sucrose-based rocket candy, but is easier to cast, less hygroscopic and does not caramelize.[24]
Sorbitol is identified as a potential key chemical intermediate[25] for production of fuels from biomass resources. Carbohydrate fractions in biomass such as cellulose undergo sequential hydrolysis and hydrogenation in the presence of metal catalysts to produce sorbitol.[26] Complete reduction of sorbitol opens the way to alkanes, such as hexane, which can be used as a biofuel. Hydrogen required for this reaction can be produced by aqueous phase catalytic reforming of sorbitol.[27]
- 19 C6H14O6 → 13 C6H14 + 36 CO2 + 42 H2O
The above chemical reaction is exothermic, and 1.5 moles of sorbitol generate approximately 1 mole of hexane. When hydrogen is co-fed, no carbon dioxide is produced.
Sorbitol based polyols are used in the production of polyurethane foam for the construction industry.
It is also added after electroporation of yeasts in transformation protocols, allowing the cells to recover by raising the osmolarity of the medium.
Medical importance
editAldose reductase is the first enzyme in the sorbitol-aldose reductase pathway[28] responsible for the reduction of glucose to sorbitol, as well as the reduction of galactose to galactitol. Too much sorbitol trapped in retinal cells, the cells of the lens, and the Schwann cells that myelinate peripheral nerves, is a frequent result of long-term hyperglycemia that accompanies poorly controlled diabetes. This can damage these cells, leading to retinopathy, cataracts and peripheral neuropathy, respectively.
Sorbitol is fermented in the colon and produces short-chain fatty acids, which are beneficial to overall colon health.[29]
Potential adverse effects
editSorbitol may cause allergic reactions in some people.[6] Common side effects from use as a laxative are stomach cramps, vomiting, diarrhea or rectal bleeding.[6]
Compendial status
editSee also
editReferences
edit- ^ publications.iupac.org/pac/1996/pdf/6810x1919.pdf
- ^ a b c d e Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ "Sorbitol_msds".
- ^ a b Teo G, Suzuki Y, Uratsu SL, Lampinen B, Ormonde N, Hu WK, Dejong TM, Dandekar AM (2006). "Silencing leaf sorbitol synthesis alters long-distance partitioning and apple fruit quality". Proceedings of the National Academy of Sciences of the United States of America. 103 (49): 18842–7. Bibcode:2006PNAS..10318842T. doi:10.1073/pnas.0605873103. PMC 1693749. PMID 17132742.
- ^ Kearsley, M. W.; Deis, R. C. Sorbitol and Mannitol. In Sweeteners and Sugar Alternatives in Food Technology; Ames: Oxford, 2006; pp 249-249-261.
- ^ a b c d e f g h "Sorbitol". Drugs.com. 23 November 2021. Retrieved 8 July 2022.
- ^ "Reduction of Glucose". butane.chem.uiuc.edu. Archived from the original on 2017-09-25. Retrieved 2017-10-03.
- ^ Sugar substitute
- ^ Tsuneyuki Oku and Sadako Nakamura (2002). "Digestion, absorption, fermentation, and metabolism of functional sugar substitutes and their available energy" (PDF). Pure Appl. Chem. 74 (7): 1253–1261. doi:10.1351/pac200274071253.
- ^ Campbell, Farrell (2011). Biochemistry (Seventh ed.). Brooks/Cole. ISBN 978-1-111-42564-7.
- ^ Hayes C (October 2001). "The effect of non-cariogenic sweeteners on the prevention of dental caries: a review of the evidence". Journal of Dental Education. 65 (10): 1106–1109. doi:10.1002/j.0022-0337.2001.65.10.tb03457.x. ISSN 0022-0337. PMID 11699985.
- ^ Nicolas GG, Lavoie MC (January 2011). "[Streptococcus mutans and oral streptococci in dental plaque]". Canadian Journal of Microbiology. 57 (1): 1–20. doi:10.1139/w10-095. ISSN 1480-3275. PMID 21217792.
- ^ Nelson, Cox (2005). Lehninger Principles of Biochemistry (Fourth ed.). New York: W. H. Freeman. ISBN 0-7167-4339-6.
- ^ "sorbitol". Cancer Drug Guide. American Cancer Society. Archived from the original on 2007-06-30.
- ^ Lederle FA (1995). "Epidemiology of constipation in elderly patients. Drug utilisation and cost-containment strategies". Drugs & Aging. 6 (6): 465–9. doi:10.2165/00002512-199506060-00006. PMID 7663066. S2CID 43386314.
- ^ Wells JG, Davis BR, Wachsmuth IK, et al. (September 1983). "Laboratory investigation of hemorrhagic colitis outbreaks associated with a rare Escherichia coli serotype". Journal of Clinical Microbiology. 18 (3): 512–20. doi:10.1128/JCM.18.3.512-520.1983. PMC 270845. PMID 6355145.
The organism does not ferment sorbitol; whereas 93% of E. coli of human origin are sorbitol positive
- ^ Rugolotto S, Gruber M, Solano PD, Chini L, Gobbo S, Pecori S (April 2007). "Necrotizing enterocolitis in a 850 gram infant receiving sorbitol-free sodium polystyrene sulfonate (Kayexalate): clinical and histopathologic findings". J Perinatol. 27 (4): 247–9. doi:10.1038/sj.jp.7211677. PMID 17377608.
- ^ "Kayexalate (sodium polystyrene sulfonate) powder". fda.gov. Retrieved 12 July 2015.
- ^ "Home – Catalent". catalent.com. Retrieved 12 July 2015.
- ^ "Sorbitol 70%". bttcogroup.in. Archived from the original on 10 July 2020. Retrieved 12 July 2015.
- ^ Medina J, Garrote R (2002). "The effect of two cryoprotectant mixtures on frozen surubí surimi". Brazilian Journal of Chemical Engineering. 19 (4): 419–424. doi:10.1590/S0104-66322002000400010. ISSN 0104-6632.
- ^ "Gallaher Group". gallaher-group.com. Archived from the original on 27 December 2008. Retrieved 12 July 2015.
- ^ a b Chemical and functional properties of food saccharides. Tomasik, Piotr. Boca Raton: CRC Press. 2004. ISBN 9780203495728. OCLC 317752036.
{{cite book}}: CS1 maint: others (link) - ^ Nakka R. "KNSB Propellant". nakka-rocketry.net. Retrieved 12 July 2015.
- ^ Metzger JO (2006). "Production of Liquid Hydrocarbons from Biomass". Angewandte Chemie International Edition. 45 (5): 696–698. doi:10.1002/anie.200502895. PMID 16374789.
- ^ Shrotri A, Tanksale, Akshat, Beltramini, Jorge Norberto, Gurav, Hanmant, Chilukuri, Satyanarayana V. (2012). "Conversion of cellulose to polyols over promoted nickel catalysts". Catalysis Science & Technology. 2 (9): 1852–1858. doi:10.1039/C2CY20119D.
- ^ Tanksale A, Beltramini, Jorge Norberto, Lu, GaoQing Max (2010). "A review of catalytic hydrogen production processes from biomass". Renewable and Sustainable Energy Reviews. 14 (1): 166–182. Bibcode:2010RSERv..14..166T. doi:10.1016/j.rser.2009.08.010.
- ^ Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, Yorek MA, Beebe D, et al. (2000). "Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage". Nature. 404 (6779): 787–90. Bibcode:2000Natur.404..787N. doi:10.1038/35008121. PMID 10783895. S2CID 4426750.
- ^ Islam MS, Sakaguchi E (2006). "Sorbitol-based osmotic diarrhea: Possible causes and mechanism of prevention investigated in rats". World Journal of Gastroenterology. 12 (47): 7635–7641. doi:10.3748/wjg.v12.i47.7635. PMC 4088045. PMID 17171792.
- ^ The United States Pharmacopeial Convention. "Revisions to FCC, First Supplement". Archived from the original on 5 July 2010. Retrieved 6 July 2009.
- ^ Sigma Aldrich. "D-Sorbitol". Retrieved 15 February 2022.
- ^ European Pharmacopoeia. "Index, Ph Eur" (PDF). Archived from the original (PDF) on 20 July 2011. Retrieved 6 July 2009.
- ^ British Pharmacopoeia (2009). "Index, BP 2009" (PDF). Archived from the original (PDF) on 11 April 2009. Retrieved 6 July 2009.
- ^ National Institute of Health Sciences (2016). "The Japanese Pharmacopoeia, Seventeenth Edition" (PDF). Archived from the original (PDF) on 4 March 2018. Retrieved 17 August 2018.
External links
edit- Media related to Sorbitol at Wikimedia Commons
- ^ Beebe JA, Frey PA (1998-10-01). "Galactose Mutarotase: Purification, Characterization, and Investigations of Two Important Histidine Residues". Biochemistry. 37 (42): 14989–14997. doi:10.1021/bi9816047. ISSN 0006-2960.

