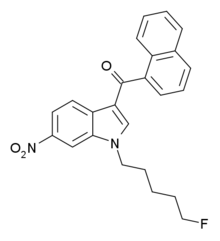
AM-1235 (1-(5-fluoropentyl)-3-(naphthalen-1-oyl)-6-nitroindole) is a drug that acts as a potent and reasonably selective agonist for the cannabinoid receptor CB1.
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C24H21FN2O3 |
| Molar mass | 404.441 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Pharmacology
editPharmacodynamics
editAM-1235 is a cannabinoid receptor agonist with Ki of 1.5 nM at CB1 compared to 20.4 nM at CB2.[1] While the 6-nitro substitution on the indole ring reduces affinity for both CB1 and CB2 relative to the unsubstituted parent compound AM-2201, CB2 affinity is reduced much more, resulting in a CB1 selectivity of around 13 times.[2] This is in contrast to other related compounds such as AM-1221 where a 6-nitro substitution instead confers significant selectivity for CB2.[3]
Pharmacokinetics
editThis section needs additional citations for verification. (July 2012) |
AM-1235 metabolism differs only slightly from that of JWH-018. AM-1235 N-dealkylation produces fluoropentane instead of pentane (or plain alkanes in general). It has been speculated that the fluoropentane might function as an alkylating agent or is further metabolized into toxic fluoroacetic acid. This is not true since fluoroalkanes do not act as alkylating agents under normal conditions and uneven fluoroalkane chains metabolize into substantially less toxic fluoropropanoic acid.[4][5]
Legal status
editIn the United States, all CB1 receptor agonists of the 3-(1-naphthoyl)indole class such as AM-1235 are Schedule I Controlled Substances.[6]
See also
editReferences
edit- ^ US granted 7241799, Makriyannis A, Deng H, "Cannabimimetic indole derivatives", published 2004-11-05, issued 2007-07-10
- ^ Deng H (2000). Design and synthesis of selective cannabinoid receptor ligands: Aminoalkylindole and other heterocyclic analogs (PhD. Dissertation). University of Connecticut. ProQuest 304624325.
- ^ WO granted 200128557, Makriyannis A, Deng H, "Cannabimimetic indole derivatives", published 2001-04-26, issued 2001-06-07
- ^ Millington JE, Pattison FL (November 1956). "Toxic fluorine compounds: XII. Esters of ω-fluoroalcohols". Canadian Journal of Chemistry. 34 (11): 1532–1541. doi:10.1139/v56-200.
- ^ Pattison FL, Howell WC, Woolford RG (February 1957). "Toxic fluorine compounds: XIII. ω-Fluoroalkyl ethers". Canadian Journal of Chemistry. 35 (2): 141–148. doi:10.1139/v57-021.
- ^ : Schedules of controlled substances
